Received: Wed 27, Nov 2024
Accepted: Mon 16, Dec 2024
Abstract
Surface osteosarcomas are rare subtypes of osteosarcoma characterized by their development on the surface of cortical bone. According to WHO 2020 classification, surface osteosarcomas include three entities:
• Parosteal osteosarcoma, periosteal osteosarcoma and high-grade surface osteosarcoma. Parosteal osteosarcoma (POS), is the most frequent subtype. It originates from the outer fibrous layer of the periosteum. The classic parosteal osteosarcoma (cPOS) has a low grade of malignancy and is mainly fibroblastic. It sometimes progresses to a high-grade osteosarcoma via dedifferentiation. Dedifferentiated POS (DPOS) has a high propensity to metastasize.
• Periosteal osteosarcoma (PerOS) originates from the germinal inner layer of the periosteum. It is mostly chondroblastic and has an intermediate grade of malignancy.
• High-grade surface osteosarcoma (HGSO) is the rarest subtype. It includes all osteosarcomas located on the surface of the bone and that are of high grade of malignancy. It originates either from the inner layer or the outer layer of the periosteum. Unlike POS or PerOS, HGSO doesn't have specific radiographic or histological features.
We propose a reclassification of the described lesions based mainly on their origin which is determinant for their radiological pattern. Therefore, there are actually only two subtypes: periosteal and parosteal OS.
Keywords
Parosteal osteosarcoma, periosteal osteosarcoma, high-grade surface osteosarcoma, classification
1. Introduction
Osteosarcoma is a primary bone malignancy characterized by malignant cells of mesenchymal origin depositing immature osteoid matrix [1]. The spectrum of lesions is very wide; they can be distinguished either by their grade of malignancy, the differentiation of neoplastic cells or even according to their location in the bone. A few entities arise on the cortical surface thereby they are called surface osteosarcomas. Surface osteosarcomas are subdivided into three patterns: Parosteal osteosarcoma (POS), periosteal osteosarcoma (PerOS) and high-grade surface osteosarcoma (HGSO) [2]. They represent respectively 5%, 1.5% and 0.5% of all osteosarcomas [1, 3]. POS is characterized by a low grade of anaplasia. PerOS has an intermediate histological grade of malignancy. HGSO is an entirely high-grade tumor.
Surgical resection with wide margins is the cornerstone of the treatment of surface OS. These lesions are less sensitive to chemotherapy than conventional high-grade OS. It’s advisable to use chemotherapy only for high-grade lesions as an adjuvant treatment. Low and intermediate grade lesions have a good prognosis with up to 80% five-year survival rate. However, high-grade lesions have a low survival rate [4].
2. Pathogenesis of Surface OS
Understanding the histology of the periosteum helps decode the pathogenesis of surface OS. The periosteum consists of two layers [5].
• The outer fibrous membrane: composed of mostly collagenous hypocellular connective tissue.
• The inner cellular layer: also called the cambium layer, made up of osteoprogenitor cells which are multipotent stem cells that can undergo mitotic division and differentiate into osteoblasts. This layer is a highly vascularized structure and it’s known as the osteogenetic layer.
POS originates from the outer fibrous layer. However, PerOS and HGSO originate from the inner cellular layer. This is consistent with radiological and pathological features as we’ll see below.
3. Parosteal Osteosarcoma (POS)
3.1. Epidemiology
POS is the most frequent surface osteosarcoma subtype but it remains rare representing less than 5% of all osteosarcoma [6, 7]. POS occurs in young adults with a mean age of about 25 years [7, 8] which differentiates it from the common form that occurs in the second decade of life. It affects both sexes with a slight female predominance [6].
The low-grade well differentiated POS is the classic type (cPOS) and the most common one. Sometimes, cPOS gets dedifferentiated and develops more aggressive behavior. It’s then called dedifferentiated POS (DPOS) [9]. Actually, the real incidence of dedifferentiation is underestimated since some lesions seen at an advanced stage are misdiagnosed as conventional OS or confused with HGSO.
3.2. Localization
POS has a predilection for the posterior aspect of the distal femoral metaphysis. The 2 main other locations are the proximal tibia and proximal humerus. Diaphyseal location is possible but uncommon [6, 8, 10, 11].
3.3. Clinical Findings
cPOS is a very slow growing lesion. Patients usually complain of long history of painless lump or an articular discomfort related to a joint stiffness, mainly knee flexion contracture. The tumor can even be diagnosed incidentally [6]. When the tumor is more symptomatic with a painful rapid growing mass, dedifferentiation must be suspected.
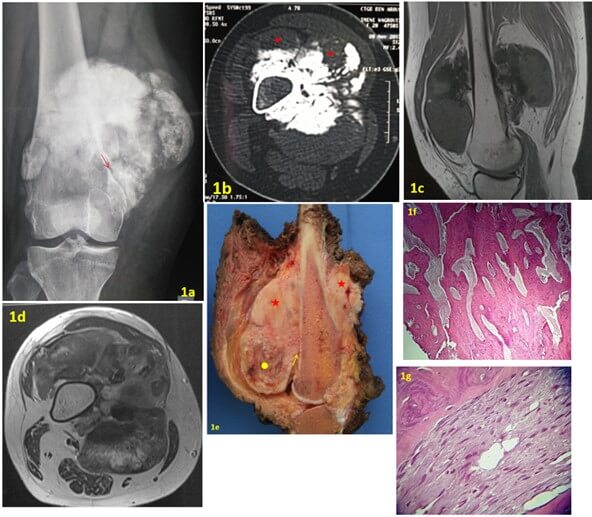
FIGURE 1: a) AP X-ray of the right knee shows an ossified tumor on the surface of the femur. A radiolucent cleavage plane (arrow) is seen between the tumor and the underlying cortex. b) Transversal CT image showing an ossified lesion developed at the surface of bone cortex. Some areas are not ossified (red point). c & d) MR Images showing a broad implantation base of the tumor and the absence of medullary extension. e) Gross specimen showing a large mass pasted on the underlying cortex with no medullary invasion. The cut surface displayed a heterogeneous appearance with ossified (red stars) and flechy areas (yellow circle). A thin lucent line (yellow arrow) is seen between the tumor and bone corresponding to the radiolucent ckeavage plane seen on X-ray. f) Photomicrograph showing an extensive bone formation arranged in trabecular streamers with fibroblastic stroma (original magnification x 40; H-E stain). g) The tumor is consisting of spindle-cell with minimal atypia between the bone trabeculae (original magnification x 125; H-E stain).
3.4. Radiological Features
On the X-ray, POS classically appears as a “melon-shaped” mineralized tumor pasted on the cortex of the host bone. It often manifests as a large mass of about 5 to 10 cm or even larger with a broad base and a lobular morphology [8]. The tumor is typically separated from the bone by a radiolucent cleavage space [2] representing the interposition of the periosteum between the cortex and the mass [12] (Figures 1a & 2a). CT scan confirms the surface ossified mass and is useful to detect cortical involvement and presence of medullary invasion (Figure 1b). MRI is important to assess local extension and medullary involvement (Figures 1c, 2b & 2c).
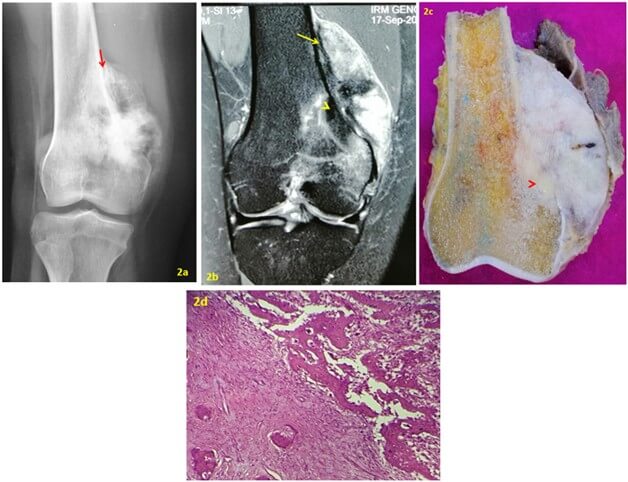
FIGURE 2: a) AP X-ray of the right knee shows an ossified peripheral tumor in the distal metaphysis of the femur with a lucent cleavage plane at its edge (red arrow). b) MRI coronal DP fat sat. The tumor is developed on the surface of the periosteum (yellow arrow). There’s a focal intramedullary extension (arrow head). c) Gross specimen showing a large, gritty white mass pasted on the underlying cortex with medullary invasion (arrow head). d) Photomicrograph showing an area of low-grade paraosteal osteosarcoma (left side) adjacent to an area of conventional high-grade osteosarcoma with the fine lacelike osteoid and pleomorphic spindle-cell. The two components are separated by a sharp transition zone (original magnification, x 80; H-E stain).
Medullary invasion is correlated with grade of malignancy for some authors [9, 13] and can be therefore a distinctive feature between cPOS and DPOS. We have reported in a previous study that invasion of the medullary canal was more frequent in DPOS than in cPOS (100% vs 25%) but it is debatable [2]. Bertoni [9] studied the meaning of radiolucencies in parosteal osteosarcomas and concluded that the presence of deep intralesional radiolucencies within a parosteal osteosarcoma strongly suggests possible dedifferentiation.
3.5. Histology
In macroscopy, POS appears as a hard ossified and lobulated mass arising from the cortical surface of the bone and attached to it with a broad base (Figures 1e & 2d). The cleavage space separating the lobulated mass from the periosteum which is seen on X-rays attests to its outer layer origin [6]. Microscopically, the cPOS is composed of mature trabecular bone associated to a fibroblastic spindle cell stroma [4]. This tissue has moderate cellularity and shows minimum cytological atypia and rare mitotic activity (Figures 1f & 1g). The presence of a component of higher grade in POS attests of dedifferentiation (Figure 2e). The high-grade component can be fibroblastic, osteoblastic or chondroblastic.
3.6. Genetics
POS is characterized by a high rate of MDM2 amplification (chromosome 12q13-15) in up to 83% of tumors [14, 15]. CDK4 gene is located in the same region and is often co-amplified and over expressed in 67% of POS. As the percentage of CDK4 and MDM2 amplification in POS is much higher than in conventional OS, most likely the MDM2/CDK4 amplified high-grade tumors represent a progression from a low-grade POS.
3.7. Differential Diagnosis
On X-rays the POS is frequently confused with other ossified bone surface lesions especially myositis ossificans and osteochondroma [16]. Upon histological examination, cPOS with its well differentiated and parallel bone trabeculae is classically overlapped with a callus of fracture, fibrous dysplasia or Paget’s disease [5]. Discrimination of DPOS from conventional high-grade OS in the absence of cPOS features is challenging especially when there is involvement of the medullary canal. In these cases, immuno-histochemistry (IHC) can be helpful with the use of MDM2 antibodies. Since conventional OS is mostly negative to MDM2, positive tumors can be considered as a progression from a cPOS. FISH for MDM2 amplification is more sensitive and specific than IHC [17].
3.8. Prognosis
cPOS has a good prognosis. Survival rate is as high as 100% when tumor is resected with safe margins [8, 9]. Local recurrences are usually secondary to intralesional or marginal resection and metastases are exceptional (less than 5%) [9]. The most important risk of cPOS is progression in the grade of malignancy called dedifferentiation which can be detected either at the time of the initial diagnosis (synchronous: 2/3 of cases) or at the time of the recurrence (metachronous) [9]. The risk of dedifferentiation is not well evaluated in the literature. Jelinek et al. [18] analyzed sixty parosteal osteosarcomas and they found that between 22% and 64% of parosteal osteosarcomas may contain components with a higher degree of analpasia.
In patients with DPOS, the metastatic rate is much higher (30-50 %) [9]. Survival data for DPOS are discordant in different series. Some studies have demonstrated poor overall survival [19], while other studies have shown a better prognosis [9].
3.9. Treatment
Surgery is the main treatment of cPOS. In cases without medullary involvement, unicortical tumor resection with marginal margins can be a safe procedure when it is feasible [1, 20]. However, DPOS needs to be treated more aggressively. Wide margins are mandatory to avoid local recurrence. Taking into account the high metastatic risk, chemotherapy is indicated. However, its efficacy is debatable. Sheth et al. [20] found that neoadjuvant chemotherapy may improve the clinical outcome. In other studies the clinical importance of chemotherapy associated with wide surgical resection is not clear [21]. Due to the poor response to chemotherapy which associated with a high risk of tumor progression, it is recommended to use chemotherapy as adjuvant after surgery for DPOS [4].
4. Periosteal Osteosarcoma
4.1. Definition
Periosteal osteosarcoma (PerOS) is an intermediate-grade OS mainly chondroblastic, arising on the surface of the bone and originating from the inner germinative layer of the periosteum [22]. Although the lesion was first recognized by Ewing in 1939, the term “Periosteal Osteogenic Sarcoma” and its distinct clinicopathological description was given by Unni et al. in 1976 [23-25].
4.2. Epidemiology
This rare tumor represents less than 2% of all osteosarcomas [28]. It occurs slightly higher in males and in younger patients compared to POS [22-27].
4.3. Localization
This neoplasm is usually located in long bones. Its preferred sites are the tibia and the femur. It evolves in the diaphysis and has rarely a metaphyseal location [28].
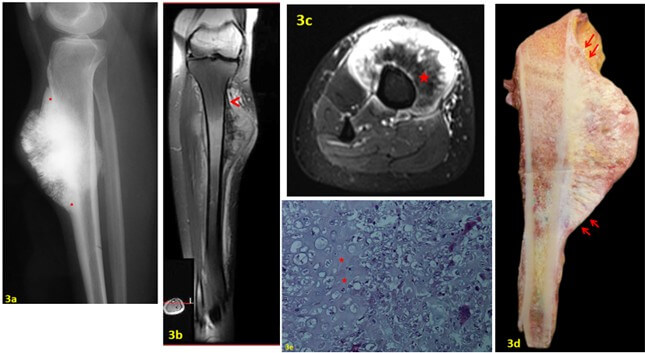
FIGURE 3: a) Lateral view X-ray of the leg showing an ossified peripheral mass on the tibial shaft with a thickening at the edge (red point) and a perpendicular periosteal reaction in the center. b & c) T1 FAT SAT after Gado MRI showing that the mass is developing under the periosteum (arrow head) that lifts it. Note the absence of medullary extension. The hypo-intensity signal (red star) indicates the cartilaginous matrix. d) The patient had wide resection. Note that the tumor develops under the periosteum (red arrows) and the medullary canal is tumor free. e) Lower-power photomicrograph showing a periosteal osteosarcoma. The tumor is predominantly cartilaginous with lace-like osteoid production (red stars) (hematoxylin and eosin,40).
4.4. Symptoms
The prominent clinical symptom is swelling. Growth rate is usually slow. The pain, when present, is generally moderate [28].
4.5. Radiological Features
In radiography, PerOS has typically a saucer shape appearance. We observe a broad-based fusiform soft tissue mass. The cortex is thickened at the periphery by a solid or lamellated periosteal reaction with occasionally a Codman triangle [4, 29]. In the center, there is a scalloping of the cortex associated to periosteal spicules perpendicular to the long axis of the shaft (Figure 3a). The CT is useful to focus on the cortex erosion and the periosteal reaction. MRI is important to detect medullary involvement and to evaluate soft tissue invasion [28, 29]. We can easily see the periosteum covering the soft tissue mass which is consistent with the origin of this tumor (Figure 3b).
4.6. Histology
Macroscopically, PerOS has a small to moderate size and its outer surface is well demarcated from the soft tissues by the periosteum [1, 28] (Figure 3d). The typical histological feature is large irregular lobules of chondroblastic tissue with small areas of osteoid formation (Figure 3e). The tumor is graded 2 or 3 when it is chondroblastic and at least 2 in osteoblastic or fibroblastic differentiation [22, 30].
4.7. Genetics
Recent findings have highlighted the genetic origins of PerOS. A mutation in at least one allele of the TP53 gene (exon 8) which is responsible for DNA repair and apoptosis has been found. This mutation leads to the inactivation of this gene via MDM2 protein regulation which directs it to the ubiquitin-mediated degradation pathway which is in turn linked to Wnt signaling [28].
4.8. Differential Diagnosis
• HGOS: It has a more aggressive course. Tumor cells are grade 3 or 4 in Broder’s classification. However, in chondroblastic differentiation, only grade 4 tumors are considered as HGSO [1, 19].
• Periosteal chondrosarcoma: The location is different and is usually metaphyseal. It is composed of malignant cartilage without any osteoid producing cells [31].
• POS: Imagery is determinant for making the difference between the two entities [1].
4.9. Prognosis
Cesari et al. [22] reported a 10YOS rate of PerOS of 84% indicating that this tumor has a better prognosis then HGSO but worse than POS. PerOS has a low propensity to metastasize; however inadequate surgical margins (marginal or intralesional) lead to local recurrence [1].
4.10. Treatment
The treatment consists of wide resection to achieve safe margins. In literature, some authors opted for a partial resection of the cortex for small limited lesions [4]. Amputation can be discussed for expanded tumors or in case of recurrence with medullar involvement [28]. Chemotherapy has been occasionally prescribed but has not shown any benefit in this intermediate grade OS [22].
5. High-Grade Surface Osteosarcoma
5.1. Definition
HGSO is the rarest subtype of surface osteosarcomas [19, 32]. This lesion was first described by Francis et al. [33] in 1964 but it was not until 1984 that Wold et al. [34] reported the first series of this extremely rare tumor. HGSO originates either from the inner or the outer layer of periosteum. Apart from its surface location, it does not differ histologically from conventional intramedullary osteosarcomas [34].
5.2. Epidemiology
HGSO accounts for less than 0.5% of all osteosarcomas. Here also, we think that the real number of HGSO is underestimated, since cases that progressed largely in the medullary canal are undistinguishable from conventional high-grade OS. The mean age of onset is around the second or the third decade and men are affected more frequently than women [19].
5.3. Localization
The most frequently involved sites are those of POS and PerOS, namely the midshaft of the femur and the tibia and the distal femur [35, 36, 37].
5.4. Symptoms
HGSO is more aggressive than other patterns of surface osteosarcomas leading to rapid progression of the disease. The mean duration of symptoms is 6 months [36]. Pain and swelling are the most frequent symptoms.
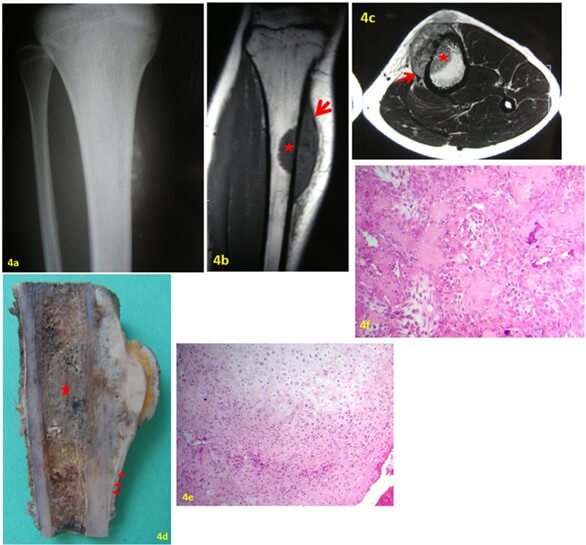
FIGURE 4: a) AP X-ray of the right tibia: soft tissue mass on the surface of the tibia moderately mineralized giving a fluffy immature appearance. b & c) MRI showing that the tumor mostly developed on the bone surface with focal medullary canal extension (star). Note that the tumor is lifting the periosteum from the cortical bone (red arrows). d) Gross specimen showing a white to grayish tumor that develops in the deeper part of the periosteum and shows lifting of the periosteum (red arrows). Large bone marrow invasion (red star). e & d) HGSO with predominant chondroblastic differentiation.
5.5. Imaging
HGSO is described in the literature [19] as a dense to moderate mineralization with a fluffy immature appearance (Figure 4a). In cross-sectional studies, the tumor usually involves on average half of the circumference of the hosting bone (Figure 4b). MRI is very helpful to show the lifting of the periosteum by the tumor (Figures 4b & 4c). An extension to the medullary canal is observed in about half of the cases [29] (Figures 4b & 4c). In these cases, involvement of the medullary canal should be limited unless classic high-grade OS is considered.
5.6. Histological Features
Basically, the histological features are those of conventional OS. The tumor exhibits frankly malignant spindle cells with atypia graded 3 or 4 according to Broder’s classification (Figure 4e). Tumor cell differentiation is either osteoblastic, fibroblastic or chondroblastic (Figure 4f) [9, 38]. In the latter form, tumor cells should be graded 4 unless the tumor is classified as PerOS.
5.7. Genetics
The genetic alterations observed in HGSO are very heterogeneous and findings have been largely inconsistent [15].
5.8. Prognosis
HGSO has the worst prognosis of the three subtypes, even worse than conventional intramedullary OS. The 5-year survival rate was reported to be as low as 37.6% [36]. One of the reasons for this poor survival rate is the high propensity of this tumor to metastasize [1, 36]. Deng [36] reported a rate of metastases of 70%.
5.9. Treatment
Surgery is the cornerstone of the treatment of HGSO. Resection of the tumor with wide margins is necessary to avoid local recurrence [36]. Even though clinical studies have failed to demonstrate any benefit of chemotherapy either in disease free survival or in overall survival [37], systemic therapy is recommended due to the high risk of distant metastasis in HGSO. The chemotherapy regimen is similar to that of conventional OS and it’s administered as adjuvant to surgery [39].
6. Reclassification of the Lesions
HGSO seems to be the least defined lesion of the three subtypes. It includes all the tumors that are of a high grade and are located on the surface of bone regardless of their:
• Clinical Behavior: Some patients included in the series of Stall [37] had a duration of symptoms of 10 years which is inconsistent with a high grade sarcoma.
• Origin: HGSO can originate either from the inner layer or the outer layer of the periosteum [31].
• Radiological Appearance: HGSO doesn’t have proper radiographic features. Cases illustrated in the literature [31, 35-37] are highly evocative of either POS or PerOS.
• Histological Differentiation
We think that the current classification of HGSO includes a certain number of DPOS with a misdiagnosed cPOS component. Indeed, some metachronous DPOS are entirely of high grade. Based on the origin of the tumor and hens its radiological appearance, we think that there are actually only two entities of surface osteosarcomas:
• In one hand, we have tumors originating from the outer layer of periosteum called the parosteal osteosarcomas. They are grossly recognized on imaging as a “melon shaped” ossified mass pasted on the bone surface and separated from it by a radiolucent line (Figure 5). With the progression of the disease, the tumor can lose some of these radiographic features and exhibit more aggressive behavior according to its grade of malignancy. Histologically, the cPOS is a low-grade tumor and the DPOS is a high-grade one. These tumors are MDM2 and CDK4 positive.
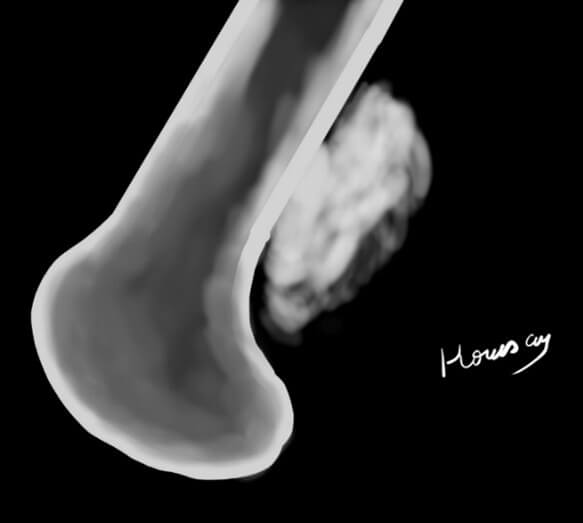
• In the other hand, we have tumors originating from the inner layer of the periosteum. They are characterized radiologically by a “saucer shape” appearance (Figure 6) and histologically, they are mainly chondroblastic. The periosteal OS is the intermediate grade and the HGSO is high grade counterpart. Future studies will define the relationship between these two entities.

Thus, we have excluded from the group of HGSO, tumors that are originating from the outer layer which are actually DPOS. Discriminative features of these two tumors are mainly radiological. In litigious cases, the study of MDM2/CDK4 amplification can be helpful. Anyway, the discrimination between DPOS and HGSO is not clinically relevant since the treatment and the prognosis are the same.
Based on the origin of the tumor, we can propose a classification of surface OS as follows (Table 1).
Table 1.
Reclassification of surface osteosarcomas according to their origin.
|
Origin |
|||||
|
Inner layer |
Outer layer |
||||
|
Grade of malignancy |
Low or intermediate |
PerOS |
cPOS |
Wide resection |
Treatment |
|
High |
HGSO |
DPOS |
Wide resection + Adj CT |
||
|
Saucer shape Located under the periosteum |
Melon shape radiolucent line MDM2/CDK4 + |
||||
|
Distinctive features |
|||||
REFERENCES
1. Venkatesan
Sampath Kumar, Nilesh Barwar, Shah Alam Khan “Surface osteosarcomas: Diagnosis,
treatment and outcome.” Indian J Orthop, vol. 48, no. 3, pp. 255-261,
2014. View at: Publisher Site | PubMed
2. WHO
Classification of Tumours Editorial Board “WHO Classification of Tumours of
Soft Tissue and Bone.” 5th ed. Lyon, France: IARC Press; 2020.
3. Marta
Sbaraglia, Elena Bellan, Angelo P Dei Tos “The 2020 WHO Classification of Soft
Tissue Tumours: news and perspectives.” Pathologica, vol. 113, no. 2, pp. 70-84, 2021. View at: Publisher
Site | PubMed
4. H Nouri, M Ben Maitigue, L Abid, et al.
“Surface osteosarcoma: Clinical features
and therapeutic implications. ” J Bone Oncol, vol. 4, no. 4, pp. 115-123, 2015.
View at: Publisher Site | PubMed
5. Ahmed Nahian,
Pradip R. Chauhan “Histology, Periosteum And Endosteum.” In: StatPearls
[Internet]. Treasure Island (FL): StatPearls Publishing; 2021. View at: PubMed
6. Jen-Fan Hang,
Paul Chih-Hsueh Chen “Parosteal osteosarcoma.” Arch Pathol Lab Med, vol.
138, no. 5, pp. 694-699, 2014. View at: Publisher
Site | PubMed
7. Antoine Babinet, Bernard Tomeno, Michel
Forest “Ostéosarcome de basse malignité. ” EMC, 2001.
8. Na Li, Lan Li,
Luciana La Tegola, et al. “Parosteal osteosarcoma with focal fatty metaplasia:
A case report.” Radiol Case Rep, vol. 14, no. 2, pp. 200-203, 2018. View
at: Publisher Site | PubMed
9. Franco Bertoni,
Patrizia Bacchini, Eric L Staals, et al. “Dedifferentiated parosteal
osteosarcoma: the experience of the Rizzoli Institute.” Cancer, vol.
103, no. 11, pp. 2373-2382, 2005. View at: Publisher Site | PubMed
10. Mohammed Sadiq,
Mohammed Ismail, K Dhanya, et al. “Parosteal Osteosarcoma of the Proximal
Phalanx of a Finger.” J Hand Surg Am, vol. 45, no. 7, pp. 663.e1-663.e7,
2020. View at: Publisher Site | PubMed
11. D A Kumm, J
Rütt, M H Hackenbroch “Parosteal osteosarcoma of the tarsus.” Arch Orthop
Trauma Surg, vol. 116, no. 6‑7, pp. 437-439, 1997. View at: Publisher Site | PubMed
12. Kelly Harper,
Paul Sathiadoss, Asif Saifuddin, et al. “A review of imaging of surface
sarcomas of bone.” Skeletal Radiol, vol. 50, no. 1, pp. 9-28, 2021. View
at: Publisher Site | PubMed
13. M Laitinen, M
Parry, J I Albergo, et al. “The prognostic and therapeutic factors which
influence the oncological outcome of parosteal osteosarcoma.” Bone Joint J,
vol. 97-B, no. 12, pp. 1698-1703, 2015. View at: Publisher
Site | PubMed
14. G Gamberi, P
Ragazzini, M S Benassi, et al. “Analysis of 12q13-15 Genes in Parosteal
Osteosarcoma.” Clin Orthop Relat Res, no. 377, pp. 195-204, 2000. View
at: Publisher Site | PubMed
15. Jeff W Martin,
Jeremy A Squire, Maria Zielenska “The Genetics of Osteosarcoma.” Sarcoma, vol. 2012, 627254, 2012. View at: Publisher Site | PubMed
16. Zafiria G
Papathanassiou, Marco Alberghini, Philippe Thiesse, et al. “Parosteal
osteosarcoma mimicking osteochondroma: A radio-histologic approach on two
cases.” Clin Sarcoma Res, vol. 1, no. 1, pp. 2, 2011. View at: Publisher Site | PubMed
17. Akihiko
Yoshida, Tetsuo Ushiku, Toru Motoi, et al. “Immunohistochemical analysis of
MDM2 and CDK4 distinguishes low-grade osteosarcoma from benign mimics.” Mod
Pathol, vol. 23, no. 9, pp. 1279-1288, 2010. View at: Publisher
Site | PubMed
18. J S Jelinek, M
D Murphey, M J Kransdorf, et al. “Parosteal osteosarcoma: value of MR imaging
and CT in the prediction of histologic grade.” Radiology, vol. 201, no.
3, pp. 837-842, 1996. View at: Publisher
Site | PubMed
19. K Okada, K K
Unni, R G Swee, et al. “High grade surface osteosarcoma: a clinicopathologic
study of 46 cases.” Cancer, vol. 85, no. 5, pp. 1044-1054, 1999. View
at: Publisher Site | PubMed
20. D S Sheth, A W
Yasko, A K Raymond, et al. “Conventional and dedifferentiated parosteal
osteosarcoma. Diagnosis, treatment, and outcome.” Cancer, vol. 10, no.
10, pp. 2136-2145, 1996. View at: PubMed
21. Hidetaka
Miyashita, Kazunari Yoshida, Tomoya Soma, et al. “Dedifferentiated parosteal
osteosarcoma of the maxilla: a case report and review of the literature.” J Med Case Rep, vol. 12, pp. 235, 2018. View at: Publisher
Site | PubMed
22. Marilena Cesari, Marco Alberghini,
Daniel Vanel, et al. “Periosteal osteosarcoma: A single-institution experience.” Cancer, vol. 117, no.
8, pp. 1731-1735, 2011. View at: Publisher Site | PubMed
23. Peter S Rose,
Ian D Dickey, Doris E Wenger, et al. “Periosteal osteosarcoma: long-term
outcome and risk of late recurrence.” Clin Orthop Relat Res, vol. 453,
pp. 314-317, 2006. View at: Publisher
Site | PubMed
24. K K Unni, D C
Dahlin, J W Beabout “Periosteal osteogenic sarcoma.” Cancer, vol. 37,
no. 5, pp. 2476-2485, 1976. View at: Publisher Site | PubMed
25. L LICHTENSTEIN
“Tumors of periosteal origin.” Cancer, vol. 8, no. 5, pp. 1060-1069,
1955. View at: Publisher Site | PubMed
26. Ashish Gulia,
Ajay Puri, Manish Pruthi, et al. “Oncological and functional outcome of
periosteal osteosarcoma.” Indian J Orthop, vol. 48, no. 3, pp. 279-284,
2014. View at: Publisher Site | PubMed
27. Chung Ming
Chan, Adam D Lindsay, Andre R V Spiguel, et al. “Periosteal Osteosarcoma: A
Single-Institutional Study of Factors Related to Oncologic Outcomes.” Sarcoma, vol. 2018, pp. 8631237, 2018. View at: Publisher Site | PubMed
28. Xin-Wei Liu, Ying Zi, Liang-Bi Xiang,
et al. “Periosteal
osteosarcoma: a review of clinical evidence. ” Int J Clin Exp Med, vol.
8, no. 1, pp. 37-44, 2015. View at: PubMed
29. Gail Yarmish,
Michael J Klein, Jonathan Landa, et al. “Imaging Characteristics of Primary
Osteosarcoma: Nonconventional Subtypes.” Radiographics, vol. 30, no. 6,
pp. 1653-1672, 2010. View at: Publisher Site | PubMed
30. Neelam Wadhwa
“Osteosarcoma: Diagnostic dilemmas in histopathology and prognostic factors.” Indian
J Orthop, vol. 48, no. 3, pp. 247-254, 2014. View at: Publisher
Site | PubMed
31. F Bertoni, S
Boriani, M Laus, et al. “Periosteal chondrosarcoma and periosteal osteosarcoma.
Two distinct entities.” J Bone Joint Surg Br, vol. 64, no. 3, pp.
370-376, 1982. View at: Publisher
Site | PubMed
32. Inwards CY,
Wenger D “High-Grade Surface Osteosarcoma.” In: Santini-Araujo E, Kalil RK, Bertoni
F, Park Y-K, éditeurs. Tumors and Tumor-Like Lesions of Bone: For Surgical
Pathologists, Orthopedic Surgeons and Radiologists [Internet]. London: Springer; pp. 235-240, 2015. View at: Publisher
Site
33. Francis KC,
Hutter RVP, Coley BL “Treatment of osteogenic sarcoma.” In: GT Pack, IM Ariel,
editors. Treatment of Cancer and Allied Diseases. 2nd ed. New York: Harper
& Row; vol. 8, 374-399, 1964.
34. L E Wold, K K
Unni, J W Beabout, et al. “High-grade surface osteosarcomas.” Am J Surg
Pathol, vol. 8, no. 3, pp. 181-186, 1984. View at: Publisher
Site | PubMed
35. K Okada, H
Kubota, T Ebina, et al. “High-grade surface osteosarcoma of the humerus.” Skeletal
Radiol, vol. 24, no. 7, pp. 531-534, 1995. View at: Publisher Site | PubMed
36. Zhiping Deng,
Zhen Huang, Yi Ding, et al. “High-Grade Surface Osteosarcoma: Clinical Features
and Oncologic Outcome.” J Bone Oncol, vol. 23, pp. 100288, 2020. View at: Publisher
Site | PubMed
37. Eric L Staals,
Patrizia Bacchini, Franco Bertoni “High-grade surface osteosarcoma.” Cancer,
vol. 112, no. 7, pp. 1592-1599, 2008. View at: Publisher Site | PubMed
38. Tommaso Cutilli,
Secondo Scarsella, Desiderio Di Fabio, et al. “High-grade chondroblastic and
fibroblastic osteosarcoma of the upper jaw.” Ann Maxillofac Surg, vol.
1, no. 2, pp. 176-180, 2011. View at: Publisher
Site | PubMed
39. Joseph D Lamplot, Sahitya Denduluri, Jiaqiang Qin, et al. “The Current and Future Therapies for Human Osteosarcoma.” Curr Cancer Ther Rev, vol. 9, no. 1, pp. 55-77, 2013. View at: Publisher Site | PubMed
