Received: Wed 06, Nov 2024
Accepted: Mon 25, Nov 2024
Abstract
Background: Observing patients submitted to percutaneous biopsy at our service who had post-biopsy breast magnetic resonance imaging scans, we observed tumor extension through the biopsy tract in some carcinomas, compromising skin and subcutaneous tissue. This study aimed to evaluate tumor extension through the biopsy tract in patients diagnosed with breast carcinoma undergoing neoadjuvant chemotherapy.
Methods: We evaluated tumor extension through a biopsy tract in patients included in a study protocol to assess the applicability of radioactive seed localization before breast surgery. Sixty-two patients included in the radioactive seed protocol were retrospectively evaluated. The abnormal enhancement in the biopsy tract was assessed by magnetic resonance imaging for clinical staging, with complementary target ultrasonography of the findings and ultrasound-guided biopsy of the suspected lesion.
Results: Four of the 62 patients in the protocol had abnormal enhancement in the biopsy tract. Three patients had a positive biopsy for carcinoma in the biopsy tract. The lesions that showed extension along the tract were Luminal A, Luminal B, and Luminal Hybrid carcinomas, with an average diameter of 3.9 cm.
Conclusion: Our study has shown the possibility of tumor extension through the biopsy tract in patients undergoing diagnostic percutaneous breast biopsy for breast cancer. These findings are crucial for understanding the potential risks and clinical implications of biopsy procedures.
Keywords
Breast cancer; needle biopsy; post-biopsy change; tumor prognostic factors; morphogenesis; ontogenetic anatomy
1. Background
Percutaneous breast biopsies are part of the diagnosis, staging, and therapeutic strategy of breast cancer treatment. There is no consensus on the best technique and number of specimens collected during the procedure. The needles used for biopsy vary between 8G and 16G, attached to automatic, semi-automatic, or vacuum-assisted devices. Some studies speculate that neoplastic cells could spread through the biopsy tract by cell seeding, but without scientific proof [1-3]. Observing patients referred to a post-biopsy breast magnetic resonance imaging (MRI) scan for clinical staging in our institution, we observed tumor extension through the biopsy tract in some of them, with extension to the skin and subcutaneous tissue confirmed by biopsy.
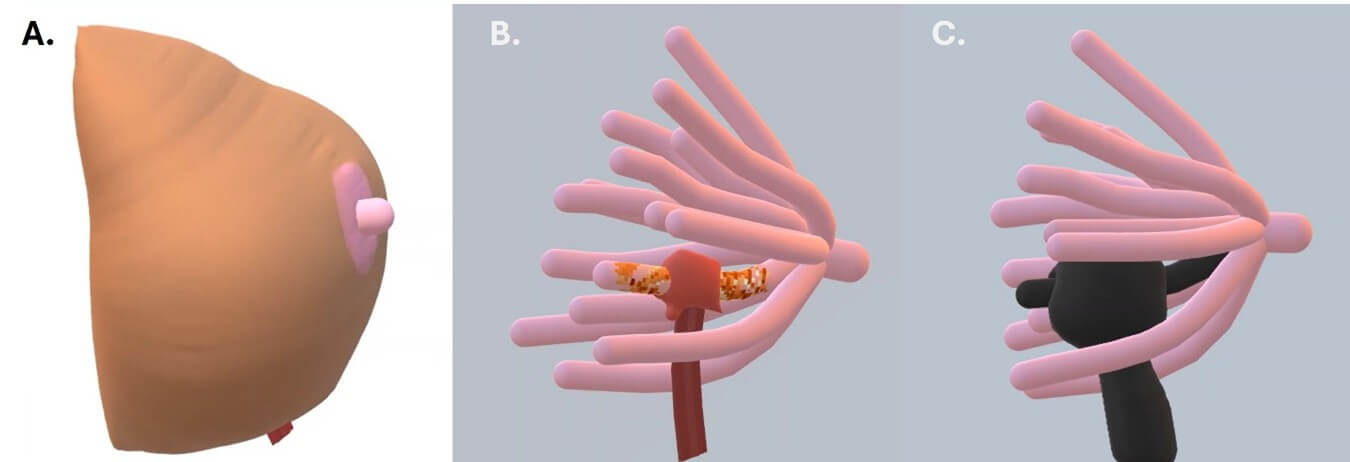
Following the theory of the ontogenetic compartment for tumor dissemination in breast cancer, the tumor should respect each duct-lobular system derived from a primordial mammary epithelium through morphogenesis. However, invasion occurs through reverse morphogenesis when the cancer spreads to a similar and adjacent ontogenetic compartment (Figure 1). Otherwise, transgression occurs when the tumor extends to a neighboring compartment of a different embryological origin. Both transgression and reverse morphogenesis are preceded by focal inflammation and fibrosis [4].
In this article, we discuss the impact of percutaneous biopsies on local tumor spread following these theories in patients included in a study protocol for radioactive seed localization before breast surgery, assuming that biopsy favors tumor cells' dissemination into the biopsy tract, which is favored by the inflammatory process inherent to the procedure. We also discuss the possible impact of breast biopsies on the tumor's microenvironment and the alternatives to improve the biopsy technique.
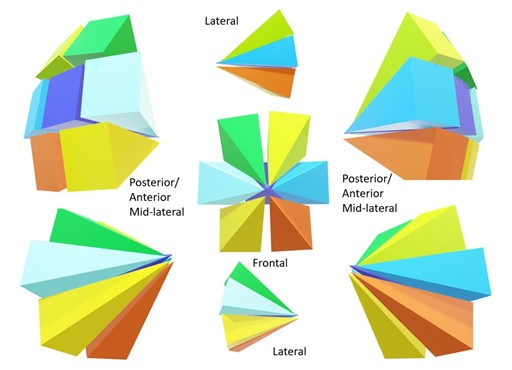
Each ontogenetic compartment is compatible with a mammary lobe, containing duct-lobular units that do not communicate with the other compartments.
2. Methods
We retrospectively evaluated MRI scans of patients included in the study protocol for evaluating radioactive seed localization before breast surgery from 01.02.2019 to 01.05.2020. The study was approved by the institutional ethics committee and, with an informed consent form signed by the patients, was registered on the Brazil Platform under "Certificado de Apresentação de Apreciação Ética" (CAAE): 45979021.9.0000.0072.
The patients included in the study were consecutively referred for radioactive seed localization before breast surgery with a diagnosis of breast carcinoma in a previous biopsy. After the radioactive localization, the patients were referred for breast MRI for preoperative clinical staging within two months of the biopsy procedure before starting neoadjuvant therapy. A radiologist with 8 years' experience in breast imaging read the MRI findings. During the interpretation process, the reader attempted to identify the site and tract of the percutaneous biopsy, from the skin and subcutaneous tissue to the periphery of the tumor. Patients whose tract was visible on MRI were recalled for targeted ultrasonography. When the tract corresponded with ultrasonography, a biopsy was performed for histopathological confirmation. The MRI protocol was a standard, using a MAGNETOM Espreee 1.5T echo scanner (Siemens, Munich, Germany) with a specific 8-channel breast coil. The contrast media (gadolinium) was administered to all patients without contraindication to the paramagnetic agent for further investigation.
Another radiologist with 20 years of experience in breast imaging performed the target ultrasonography. Ultrasonography was performed using a Toshiba Aplio 300 ultrasound system (Canon Medical Systems Corporation, Ōtawara, Tochigi, Japan) with a 5-14 mHz multifrequency linear probe. The same radiologist performed the biopsy of the suspected area using a 14g needle with a semi-automatic device BARD Core Magnum (Bard Company, New Providence, New Jersey, U.S.) with multiple insertions and at least two samples collected.
The samples were sent to the institution's Pathology Department for histological and immunohistochemical evaluation. The results were analyzed statistically using MedCalc for Windows software version 19.4 (MedCalc Software, Ostend, Belgium).
3. Results
Between 09.04.2019 and 10.06.2020, 62 patients underwent radioactive seed localization before breast surgery to mark primary breast cancer. Of these, four (6.4%) were diagnosed with a biopsy tract impairment on MRI, with three of them testing positive for malignancy. The average time between the diagnostic biopsy and the staging MRI was 45 days. Table 1 shows the characteristics of the patients and the lesions included in the protocol (Table 1). Table 2 shows the characteristics of the positive findings of the biopsy tract impairment (Table 2).
Table. 1.
Characteristics of the patients and the lesions included in the protocol.
|
Luminal A |
Luminal B |
HER2(+) |
Luminal Hybrid |
Triple Negative |
Total |
||
|
N |
9 |
21 |
4 |
12 |
16 |
62 |
|
|
(%) |
14.5% |
33.9% |
6.5% |
19.4% |
25.8% |
100.0% |
|
|
Dimension (cm) |
Median |
36.0 |
35.4 |
27.5 |
36.4 |
39.7 |
36.0 |
|
Average |
38.6 |
37.6 |
27.0 |
42.2 |
31.7 |
38.8 |
|
|
SD |
13.0 |
13.2 |
5.1 |
22.7 |
22.7 |
15.6 |
|
|
Age (year) |
Median |
52 |
|||||
|
Average |
51.2 |
||||||
|
SD |
10.5 |
||||||
Table. 2. It shows
the characteristics of the positive findings of the biopsy tract impairment.
|
Case |
Age (year) |
Dimension (cm) |
Type |
KI-67 |
|
1 |
48 |
4 |
Luminal B |
20 |
|
2 |
46 |
3.4 |
Luminal A |
5 |
|
3 |
40 |
4.1 |
Luminal Hybrid |
30 |
The average time between the diagnostic biopsy and the staging MRI was 45 days. The carcinomas diagnosed in the biopsy tracts were Luminal A, Luminal B, and Luminal Hybrid. Macrophages, lymphocytes, and giant cell granulomas with foreign body reaction were found in the negative biopsy (Figure 2).
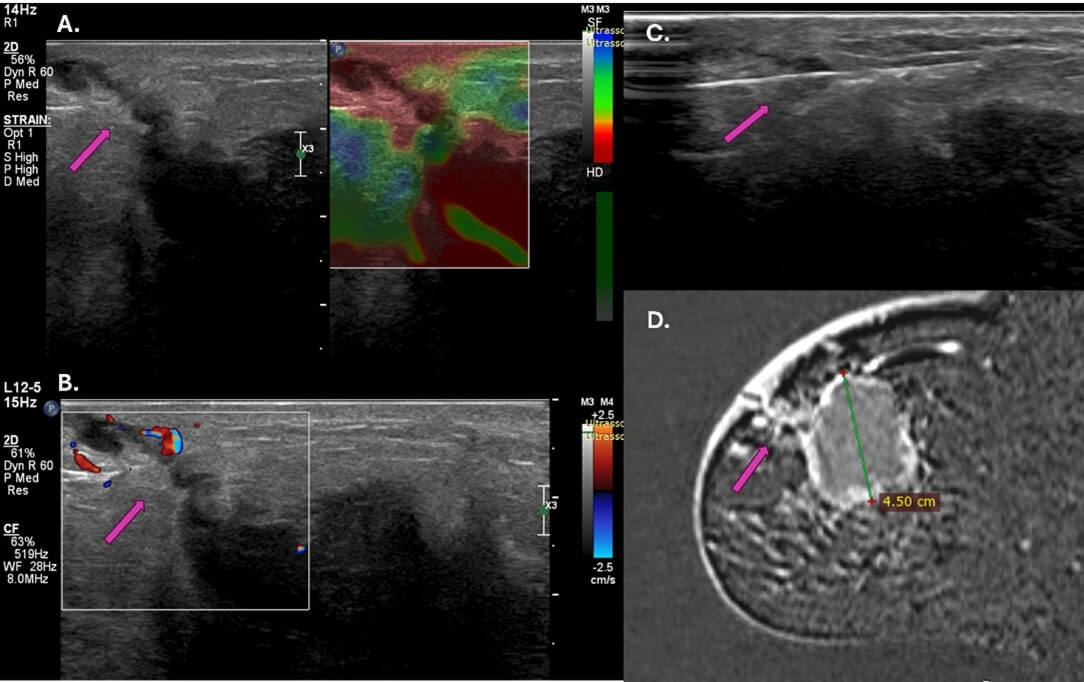
4. Discussion
Neoplastic seeding of the biopsy tract following percutaneous needle biopsy is controversial in the literature. Despite being well-documented for some types of cancer, such as hepatocellular carcinoma, non-small cell lung carcinoma, renal cell carcinoma, and thyroid carcinoma, the pathophysiology of this dissemination is not well established [1-3]. Some authors assume that the displacement of epithelial cells is responsible for this dissemination. Brenner et al. reported a 0.8% rate of neoplastic implantation [5]. Santiago et al. defined neoplastic seeding as malignant-appearing masses or calcifications that developed along the biopsy needle tract extending to subcutaneous tissues and skin following the needle biopsy, not present on imaging before or at the time of the breast biopsy. In the article published in 2017, 4010 patients diagnosed with breast cancer were retrospectively evaluated, and eight patients had a diagnosis of neoplastic seeding. The average time of appearance was 60.8 days [3].
Some concepts are essential to understanding neoplastic seeding. The female breast is formed by 15-20 lobular duct units with a shared origin in the areola-papillary complex. Embryologically, each duct-lobular unit of the breast represents a distinct sub-compartmental anatomy originating from an individual sprout of the breast epithelial primordium, denominated an ontogenetic compartment [6, 7]. An article on "Reflection and Reaction," published by Hockel in 2009, discusses the importance of the ontogenetic compartment: "For conservative resection of DCIS and early invasive cancer lesions, the margins at the circumferential border of the compartment can be close without the risk of local recurrence. Intra-compartmental margins should be substantially wider to rule out occult residual disease". In the article, the author also argues that there is clinical evidence that wound-healing reaction from cancer resection stimulates the initiation and growth of the local relapse of malignant lesions and might also enhance its malignant progression, possibly through a hypoxic environment, resulting in more aggressive disease [6].
The local dissemination of invasive carcinoma is an isotropic process of tissue infiltration independent of tumor margins, according to Hockel in another article published in 2012 [8]. The tumor microenvironment favors invasion of the interstitium, intravasation of lymphatic and venous channels, and perineural spread. The edges of the home compartment suppress tumor growth. The author stated that each breast lobe unit corresponds to a distinct ontogenetic compartment, where tumor dissemination is more favored towards this compartment's interior than the periphery. The author postulates that the invasion of a tumor into another ontogenetic compartment is favored by a local inflammatory process associated with the reverse morphogenesis or transgression of tumor cells. Functional differences are also observed between tumors extending into the same compartment and infiltrating a neighboring compartment. Tumors infiltrating the adjacent compartment are well-oxygenated, while those confined to the ontogenetic space are hypoxic [8].
Also, according to Hockel in 2019 [4], a malignant tumor originates from epigenetic alterations that increase cell proliferation, affecting the last layer of differentiated cells in an ontogenetic compartment called the "bauplan." This process is called morphogenesis. Genetic mutations associated with increased function of oncogenes and loss of function of tumor suppressor genes are known as driver mutations. The plasticity of the tumor cell, usually through a process of "backward" unfolding of the altered epigenome in malignant progression, can contribute to tumor invasion into an adjacent ontogenetic space, which can be called reverse morphogenesis. Invasion into an ontogenetic space of another origin, such as the skin and subcutaneous tissue, would be called transgression.
In 2023, McCarty et al. [9] published "Sustained Inflammation of Breast Tumors Affecting Needle Biopsy". According to the authors, performing a biopsy triggers a wound-healing response and a foreign-body reaction. Inflammation, hemorrhage, fat necrosis, granulation tissue, necrosis, and giant cell reaction are common findings in the biopsy tract, as well as atypical spindle cells and atypical duct-like structures have also been reported. The primary tumor cells in the biopsy tract have been observed in some cases. The wound-healing process consists of four phases: hemostasis, inflammation, proliferation, and remodeling. The inflammatory phase predominates neutrophils and eosinophils, followed by macrophages that release cytokines and chemokines. The authors demonstrate that the prolonged presence of macrophages and eosinophils slows down the wound healing process, regardless of the histological pattern of the tumor. In the study, the authors demonstrate the presence of macrophages in the biopsy site for an extended period (average 34+/- 28.8 days). The transition of macrophage phenotypes for tissue repair follows from M0 → M1 → M2. The M2 phenotypes involved in the wound-healing process and the tumor-associated macrophages (TAMs) are classified differently but have similar actions mediating cell proliferation and angiogenesis through cytokines, chemokines, and growth factors. As M2 TAMs are related to a worse prognosis, it would be essential to understand the M2/ M1 relationship after biopsy and the possible alterations in neighboring cells. In other words, there is ambiguous information for the defense system regarding the difference between repairing a tissue by wound healing and protecting the host from tumor dissemination [9].
Another peculiar factor described by McCarty is the presence of neutrophils, eosinophils, and plasma cells carrying biopsy marker material. Theoretically, the markers are wrapped in biocompatible, biodegradable material to prevent displacement. This degraded material found in inflammatory cells prolongs the inflammatory process. In our study, we used titanium capsules with radioactive iodine-125. The influence of these devices on the tumor microenvironment is not established [9].
We followed up with 62 consecutive patients subjected to consecutive placement of radioactive markers in the biopsy site, where three of the 62 patients (4.8%) showed tumor seeding along the biopsy tract within 2 months. The tumor dissemination seeding was higher in our study compared to Santiago et al., which may be explained by the study methodology, where the patients who underwent biopsy were followed up during the surgical planning interval in a prospective study. Another factor that may have contributed is magnetic resonance imaging for tumor evaluation, the most sensitive diagnostic method for detecting breast cancer. As the extension in the biopsy tract are new lesions and have the potential to change clinical/surgical management, associated with 4.8% of malignancy through the biopsy tract in our results, means that according to the breast imaging and reporting data system (BI-RADS) lexicon fifth edition recommendations, these findings should be classified as suspicious and included in category 4 [10].
As discussed above, the extension of tumor cells along the biopsy tract leads tumor cells into healthy adjacent ontogenetic compartments, favored by the inflammatory process resulting from the percutaneous procedure. Sometimes, tumor cells can reach the skin and subcutaneous tissue. This information should be especially relevant in the context of de-escalating surgery for breast cancer, where there could be a risk of tumor remnant after surgical treatment in the biopsy tract. The findings deserve more attention in cutaneous impairment and recurrence after surgical treatment, where breast carcinoma metastases account for 24 to 50% of cutaneous metastases (Figure 3) [11].
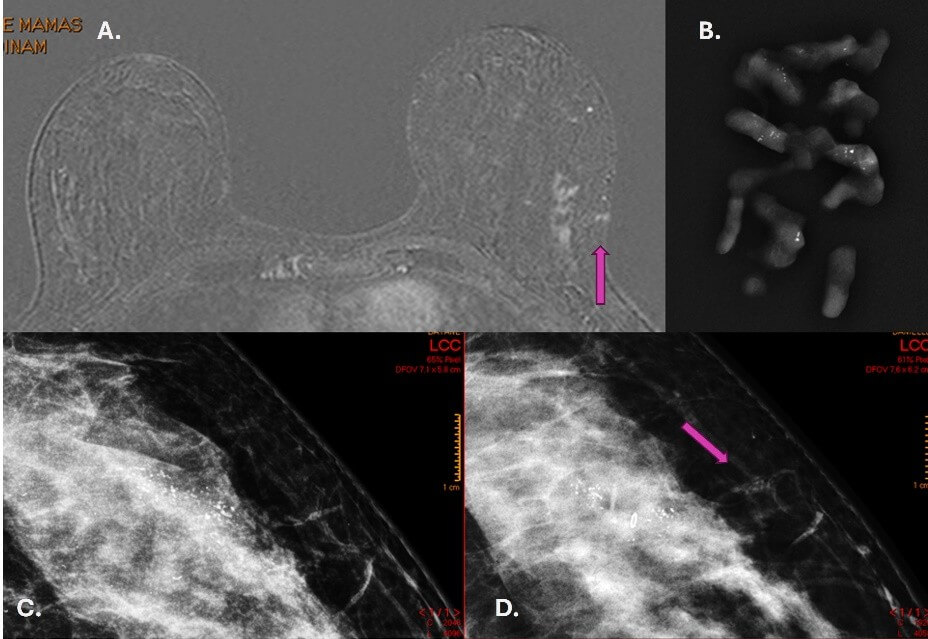
As in the studies cited here, our results showed no relationship between the immunohistochemical profile and the prevalence of tumor implantation. During the study period, we observed in our clinical practice one case of tumor implantation in pure ductal carcinoma in situ after performing a vacuum-assisted biopsy of calcifications, which corroborates our findings (Figure 4). We also demonstrated two other examples of tumor extension that were not included in our study protocol (Figure 5). Currently, we have incorporated into our practice the evaluation of the biopsy tract in patients undergoing breast carcinoma staging at magnetic resonance imaging.
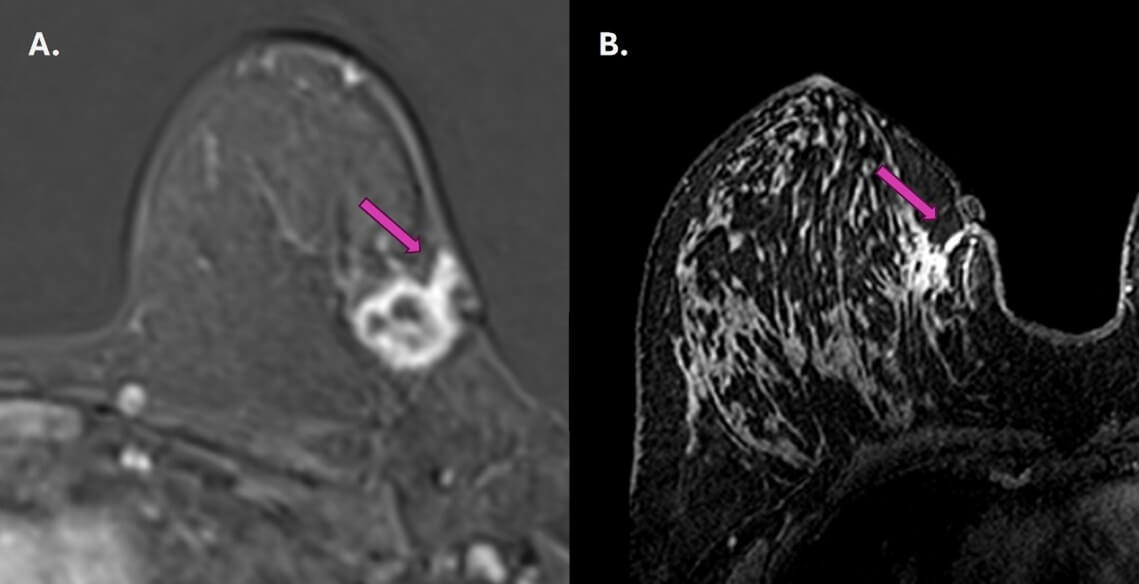
According to our findings, we believe that tumor cells seeding in the biopsy tract are a consequence of the direct extension of the primary tumor into the cavity created by the percutaneous biopsy since there is an imbalance between the defense system and the wound-healing process. McCarty et al. [9] discuss that complicated procedures with hematoma formation can delay the resolution of the wound-healing process and favor tumor extension. In this context, procedures with thinner needles, fewer fragments, and concern for adequate hemostasis with drainage of post-biopsy hematomas could minimize the chance of seeding along the biopsy tract. Other theories justify neoplastic seeding in the setting of needle biopsy, which associates epithelial cell displacement as a precursor to seeding. Liebens et al. reported displacement of malignant cells in 22% of patients who underwent large-gauge core-needle biopsy [12]. Based on this theory, the authors propose washing the procedure needles as a preventative measure to avoid cell displacement. However, according to our findings, neoplastic seeding is due to the direct infiltration of tumor cells along the biopsy tract [13, 14].
Our study has some limitations. First, it was carried out in a single institution, with only two observers. However, the study design facilitated patients' follow-up from the biopsy until the staging MRI scan. Second, due to neoadjuvant chemotherapy, the patients could only be followed up for up to 2 months, which would increase the chance of false-negative results. Prospective, multicenter studies could be carried out to confirm and replicate our findings. The presence of seeding in the biopsy tract in diagnostic biopsies in breast cancer patients is clinically relevant and can be diagnosed by tumor staging magnetic resonance imaging. It is crucial to diagnose the extent of the tumor along the biopsy tract for surgical planning and safer management of the patient.
Data Availability
All datasets generated or analyzed during the current study are available from the corresponding author on reasonable request.
Acknowledgements
None.
Ethics Approval
The study complies with the Declaration of Helsinki and was performed according to local ethics committee approval. This retrospective, single-center study, approved by the institutional ethics committee, was registered on the Brazil Platform under CAAE: 45979021.9.0000.0072.
Conflicts of Interest
None.
Funding
None.
Author Contributions
Study conception and design and material preparation were performed by EFCF. Data collection was performed by EFCF, MGMT and VJA. Data analysis was performed by EFCF, MGMT and VJA. The first draft of the manuscript was written by EFCF and revised by all authors; all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Consent to Participate
Informed consent was obtained from all individual participants included in the study.
Consent to Publish
The authors affirm that human research participants provided informed consent for publication of the images.
Abbreviations
MRI: Magnetic Resonance Imaging
TAMs: Tumor-Associated Macrophages
BI-RADS: Breast Imaging and Reporting Data System
M: Macrophage
REFERENCES
1. Arghavan Sharifi, Stephen Seiler,
Helena Hwang, et al. “Neoplastic seeding of breast cancer along the core biopsy
tract.” Breast
J,
vol. 26, no. 10, pp. 2129-2131, 2020. View at: Publisher Site | PubMed
2. Botond K Szabo, Akinyede Ojo, Dhafir
Al-Okati “MRI Diagnosis of Needle Tract Tumor Seeding Following Core Biopsy of
Mucinous Carcinoma of the Breast.” Cureus, vol. 13, no. 4, pp. e14493, 2021. View at: Publisher Site | PubMed
3. Lumarie Santiago, Beatriz E Adrada,
Monica L Huang, et al. “Breast cancer neoplastic seeding in the setting of
image-guided needle biopsies of the breast.” Breast Cancer Res Treat,
vol. 166, no. 1, pp. 29-39, 2017. View at: Publisher
Site
| PubMed
4. Michael Höckel, Ulrich Behn “The
Order of Cancer: A Theory of Malignant Progression by Inverse Morphogenesis.” Front Oncol, vol. 9, pp.
416, 2019. View at: Publisher
Site
| PubMed
5. R James Brenner, Linda M Gordon
“Malignant seeding following percutaneous breast biopsy: documentation with
comprehensive imaging and clinical implications.” Breast J, vol. 17, no.
6, pp. 651-656, 2011. View at: Publisher
Site
| PubMed
6. Michael Höckel, Nadja Dornhöfer
“Understanding and preventing local tumour recurrence.” Lancet Oncol,
vol. 10, no. 7, pp. 645-646, 2009. View at: Publisher
Site
| PubMed
7. Mariella Mannino, John Yarnold
“Effect of breast-duct anatomy and wound-healing responses on local tumour
recurrence after primary surgery for early breast cancer.” Lancet Oncol, vol. 10, no.
4, pp. 425-429, 2009. View
at: Publisher Site | PubMed
8. Michael Höckel “Cancer permeates
locally within ontogenetic compartments: clinical evidence and implications for
cancer surgery.” Future Oncol, vol. 8, no. 1, pp. 29-36, 2012. View at: Publisher Site | PubMed
9. Cruz McCarty, Misung Yi, Senna Sous,
et al. “Sustained Inflammation of Breast Tumors after Needle Biopsy.” Pathobiology,
vol. 90, no. 2, pp. 114-122, 2023. View at: Publisher Site | PubMed
10. Sickles EA, D'Orsi CJ, Bassett LW
“ACR BI-RADS mammography.” ACR BI-RADS atlas, breast imaging reporting and data
system, 5th edition. American College of Radiology, Reston, VA; pp. 134-136,
2013.
11. Samantha Huang, Vishwas Parekh,
James Waisman, et al. “Cutaneous metastasectomy: Is there a role in breast
cancer? A systematic review and overview of current treatment modalities.” J
Surg Oncol, vol. 126, no. 2, pp. 217-238, 2022. View at: Publisher Site | PubMed
12. Fabienne Liebens, Birgit Carly, Pino
Cusumano, et al. “Breast cancer seeding associated with core needle biopsies: a
systematic review.” Maturitas, vol. 62, no. 2, pp. 113-123, 2009. View
at: Publisher Site | PubMed
13. Chandandeep Nagi, Ira Bleiweiss,
Shabnam Jaffer “Epithelial displacement in breast lesions: a papillary
phenomenon.” Arch Pathol Lab Med, vol. 129, no. 11, pp. 1465-1469, 2005.
View at: Publisher Site | PubMed
14. L K Diaz, E L Wiley, L A Venta “Are malignant cells displaced by large-gauge needle core biopsy of the breast?” AJR Am J Roentgenol, vol. 173, no. 5, pp. 1303-1313, 1999. View at: Publisher Site | PubMed
