Received: Sun 15, Dec 2024
Accepted: Thu 06, Feb 2025
Abstract
Background: To evaluate population-level changes in survival of glioblastoma multiforme (GBM) and anaplastic astrocytoma (AA) between January 2011 and December 2020 in a Spanish region with 1.5 million inhabitants, and to assess whether the type of treatment applied to these tumours is reflected in population survival rates.
Methods: This study obtained data from the health records of the Region of Murcia (1.5 million inhabitants). We identified all patients (age ≥ 18 years) with a histopathological diagnosis of GMB or AA between 2011 and 2021 to calculate the incidence and mortality trends of these tumours. Patients were followed up until December 2023. Percentage results and 95% confidence intervals (95%CIs) were calculated using SPSS software (Microsoft Version 28).
Results: We identified 530 patients with GBM and 137 with AA. The mean age was 64.1 years and 44% were women. Median overall survival (OS) was 12.3 months for all patients diagnosed with GBM (95% CI: 11.3-13.8). For AA patients, the median OS was 38.4 months; 43.21 months in men (95% CI: 33.4-52.8) and 34.63 months in women (95% CI: 28.7-40.9).
Conclusion: The aggressive potential of GBM, with a one-year survival of 33%, was confirmed in this study. We have demonstrated that the presence of surgery with complete oncological treatment is the most important factor in OS. As regards sex, male patients had a longer survival, with this difference being greater in patients with AA.
Keywords
Glioma, glioblastoma, age-adjusted incidence, treatment-based mortality, survival
Key Points
• In recent years, the incidence of glioblastoma and anaplastic astrocytoma has been increasing in our region.
• Survival is greater in males, especially for anaplastic astrocytoma.
• The overall survival of both tumours in our region is equivalent to the data published in large studies, being higher if complete surgery + chemotherapy (CT) - radiotherapy (RT) is performed.
1. Importance of the Study
In Spain, primary tumours of the central nervous system (CNS) represent 2% of all cancers in adults and up to almost 15% in children under 15 years of age, with a total of 3,200 deaths per year and rising. The reasons for the increase in incidence in recent years remain unclear. There is agreement that it is partly the result of better diagnostic tests; however, improved diagnostic capacity cannot account for all this increase. These data, along with evidence suggesting that the incidence may have been growing for many decades, leave open the possibility that environmental exposure could explain some of the increasing incidence of brain tumours. Given that these tumours are extremely aggressive and have limited survival, our study aimed to discover whether this increase in incidence in recent years was real in our region and if the treatment received could affect survival for these tumours.
Furthermore, it has been known for decades that men are more likely than women to present a primary malignant brain tumour. There is also evidence that sex influences the response to treatment, although the reasons for these differences between sexes are unclear. Sex differences in cancer are an area of research that has not been sufficiently studied, and these studies are needed to advance in what is often referred to as personalised or precision medicine. Therefore, it is very important to know if these differences are real for future studies, where the ability to predict which patients, for example, are more likely to respond to treatment or undergo disease progression, could be improved.
2. Introduction
In Spain, primary central nervous system (CNS) tumours represent 2% of all cancers in adults and up to almost 15% in children under 15 years of age [1], with a total of 3,200 deaths per year [2]. Every year, 4,000 new cases of malignant gliomas are diagnosed in Spain, of which more than a third are glioblastoma multiforme (GBM). In addition, GBM has an incidence of 3-5 cases per 100,000 inhabitants [2]. Malignant gliomas account for approximately 60% of all primary brain tumours in adults.
The incidence rate in children (0-19 years) is 6.1 per 100,000 children, with a balanced sex ratio [3]. However, if we look at the percentage of malignant primary brain tumours among children and adults, it is higher in children (60% vs. 30%). The incidence is lower in children; nevertheless, brain tumours are the most common solid malignancy and the leading cause of cancer mortality in children and adolescents up to 19 years of age, followed by leukaemia and soft tissue cancer [3]. The CNS is the third most common cancer site among adolescents and young adults, and the eighth among adults aged 40 and older.
GBM can develop at all ages, with the highest incidence in the sixth decade of life and a mean age at diagnosis of 62 years. Most GBMs arise "de novo," while secondary GBMs develop from a lower-grade glioma. Approximately one-third of tumours are malignant and the rest are benign or borderline malignant [4]. In the rest of the world, the incidence is estimated at 7 per 100,000 people worldwide, which represents 2% of all primary tumours.
Since 2017, the incidence of brain tumours in Spain has increased by 12% [2, 5-7]; the reasons for this increase remain unclear. There is agreement that at least part of this increase is the result of better diagnostic tests [8, 9]. However, improved diagnostic capacity cannot explain all this increase in incidence. These data, along with evidence suggesting that the incidence may have been growing for many decades, leave open the possibility that environmental exposure could explain some of the increasing incidence of brain tumours [9].
Five-year survival rates for the most common histologic subtypes, anaplastic astrocytoma (AA) and GBM are 30% and 5.6%, respectively [3]. Overall survival (OS) in patients with malignant brain tumours has not improved significantly over the past 50 years. Nonetheless, survival rates vary according to age and histology [10]. Little progress has been made in GBM survival over the past 30 years; however, five-year survival rates for medulloblastoma increased by 20% from the 1970s to the 1980s, although data suggest a stabilisation of survival rates [11].
In general, young age, suitable functional status, and lower histopathological grade are favourable prognostic factors for primary brain tumours. Other favourable but less significant prognostic factors include the absence of changes in mental behaviour at the time of diagnosis, cerebellar location of the tumour, and complete surgical resection [12]. In terms of sex, there is a slight male predominance in the incidence of malignant brain tumours [6]. However, when evaluating the total of tumour types, both malignant and benign, men represent less than half of all cases (42% vs. 58%) [1]. This difference is mainly explained by the higher incidence of meningiomas in women.
Our study focused on analysing data regarding the most common malignant brain tumours in our setting, such as GBM (3.2 per 100,000 inhabitants) and grade 3 AA (0.51 per 100,000 inhabitants) [13].
3. Methods
3.1. Ethical Considerations
According to Spanish law, individual-level data with identifiers can only be used for scientific research with the approval of the authorities or for statistics. The data processing procedures were evaluated and approved by our hospital's ethics committee (Annexe 1) before starting our study.
3.2. Patient Selection
We retrospectively reviewed the electronic medical records of all patients diagnosed with a brain tumour with GBM or AA pathology at a tertiary care centre in Murcia, Spain, between January 2011 and December 2020. Patients were followed up until December 2023. In our study, patients presenting with GBM were considered NOS GBMs (not otherwise specified), which is a diagnosis in the current (2021) classification of CNS tumours of the World Health Organization (WHO) [14]: a diffuse glioma with astrocytic and anaplastic features, microvascular proliferation, and/or necrosis consistent with a WHO grade 4 tumour. Similarly, AA in our sample was also considered NOS. Patients with relapsed disease diagnosed before January 2011 and loss to follow-up patients were excluded from the final analysis.
3.3. Clinical and Demographic Characteristics
Clinical and demographic data were collected from the electronic medical records: age at diagnosis, type of tumour histology, sex, date of diagnosis, date of progression, date of death, treatment received, extension study before the start of treatment, cardiovascular risk factors (CVRF), active alcohol, active smoking, secondary neoplasm diagnoses, cause of death, and preoperative ECOG. OS was defined as the date from initial pathologic diagnosis to the date of death from any cause, and progression-free survival (PFS) was defined as the date from initial pathologic diagnosis to the date of radiological progression by MRI, or the date of death if there was no radiological evidence of progression.
3.4. Survival and Follow-up
Patients were followed from the date of histopathological diagnosis until death, emigration out of our system, or until the end of 2023. Due to the mandatory registers that include unique personal identifiers, the follow-up was complete.
3.5. Statistical Analysis
We calculated the standardised incidence of GBM. Incidence rates are also reported by age groups (18-30, 30-40, 40-60, and > 60 years). For all statistical methods, the result was considered significant when p < 0.05. A log-rank test was used to assess differences in OS and PFS between groups and plotted using the Kaplan-Meier method. A sub-analysis was performed in the GBM and AA groups separately via a univariate proportional hazards index model to evaluate associations between variables and OS and PFS. The specific subgroup analysis was performed by sex and treatment type. We also report average survival times. The p-values for differences in excess mortality between the two time periods were based on the likelihood ratio test, which was adjusted for multiple comparisons using the Benjamini-Hochberg procedure. Next, a multivariate proportional hazards ratio model was performed to assess the influence of each of them on our results. The log-rank test, the Kaplan-Meier curve, and Cox proportional hazards models were performed using SPSS statistical software (Microsoft Version 28) for time and the main effects of age and sex.
4. Glioblastoma Results
4.1. Patient Selection
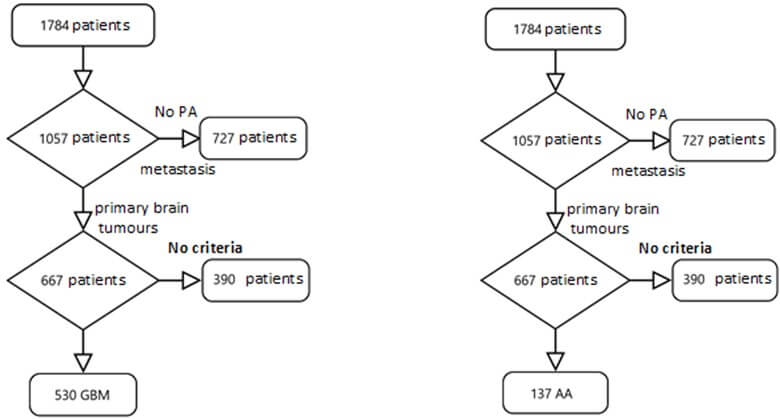
In our search, we found a total of 1784 patients, of whom there was a registry error in 727. 523 were discarded because the brain tumour was metastatic from a primary extracerebral tumour, and 205 because they lacked a pathological anatomy (PA) study as the diagnosis was made exclusively via imaging. Of the remaining 1,057 patients evaluated in the study (Table 1), 667 met the inclusion criteria for the proposed study: 530 were diagnosed with GBM and 137 with AA. The other diagnoses can be seen in (Table 1).
TABLE 1: Flow chart and diagnoses of the patients in the study.
|
Diagnosis |
Frequency |
Percentage |
|
Glioblastoma |
530 |
50.14 |
|
Anaplastic astrocytoma |
137 |
12.96 |
|
Meningiomas |
122 |
11.54 |
|
Grade II astrocytoma |
67 |
6.34 |
|
Ependymoma |
55 |
5.20 |
|
Anaplastic oligodendroglioma |
39 |
3.69 |
|
Anaplastic ependymoma |
26 |
2.46 |
|
Gliosarcoma |
18 |
1.70 |
|
Grade II oligodendroglioma |
14 |
1.32 |
|
Trunk glioma |
12 |
1.14 |
|
Medulloblastoma |
9 |
0.85 |
|
Glomus jugulare |
6 |
0.57 |
|
Pilomyxoid astrocytoma |
5 |
0.47 |
|
Gemistocytic astrocytoma |
4 |
0.38 |
|
Meningothelial meningioma |
3 |
0.28 |
|
Giant cell astrocytoma |
2 |
0.19 |
|
Esthesioneuroblastoma |
2 |
0.19 |
|
Pleomorphic xanthoastrocytoma |
2 |
0.19 |
|
Grade II pineal tumour |
2 |
0.19 |
|
Grade IV primitive
neuro-ectodermal tumour (PNETs) |
2 |
0.19 |
|
Total |
1057 |
100.0 |
If we examine GBM diagnoses per year to assess whether there has been an increase in incidence in recent years, we observe the following results:
2011: 44, 2012: 47, 2013: 43, 2014: 53, 2015: 52, 2016: 50, 2017: 52, 2018: 60, 2019: 62 and 2020: 67.
4.2. Clinical and Demographic Characteristics
The median age at diagnosis of the sample was 68.22 years, (range: 18-90, standard deviation (SD): 19.29). The median age of death was 62.35 years (range: 19-91, SD: 18.61). If we stratify GBM into age groups of 18-30, 30-40, 40-60, and > 60 years, we observe that patients over 60 years of age constituted the largest GBM population (Table 2).
TABLE 2: Percentage of patients by age range.
|
Glioblastoma Age (years) |
Percentage |
Anaplastic Astrocytoma Age (years) |
Percentage |
|
|
|
18-30 |
|
18-30 |
6.5 |
|
30-40 |
|
30-40 |
14.7 |
|
|
40-60 |
|
40-60 |
34.7 |
|
|
>60 |
|
>60 |
44.1 |
|
|
Total |
|
Total |
100 |
|
In our cohort, there was a slightly higher percentage of men than women (55.7% vs. 44.3%, respectively). Regarding patients’ functional status measured by the ECOG scale at diagnosis, 138 patients (26%) had an ECOG of 0, 322 (60.8%) had an ECOG of 1, and 70 (13.2%) had an ECOG of 2 or higher.
Fifty-eight patients (7.4%) had other primary neoplasms in addition to the primary CNS tumour, all of which were metachronous tumours. Of these 58 patients, 42 (6.3%) had a disease-free period greater than five years since the other tumour was diagnosed. None of the patients had metastatic disease of the primary non-CNS tumour.
Regarding the treatment received by patients, it is noteworthy that more than half of the patients were treated with (complete) surgery + chemotherapy (CT) + radiotherapy (RT) (58%). The second most common option was incomplete surgery/biopsy without any further treatment. Concerning the specific oncological treatment received by each patient, we highlight that only 8.68% did not receive any type of CT and that more than half of the patients received 60 Gy of brain RT treatment. The percentage of patients per treatment is specified in (Table 3).
TABLE 3: Type of treatment received by patients with glioblastomas and survival
outcomes by type of treatment.
|
Types of treatments |
Frequency |
Percentage |
Progression-free Survival (months) |
|
Overall Survival (months) |
|
|
|
|
Surgery + CT + RT |
309 |
58.30% |
9.493 |
|
18.645 |
|
|
Surgery + RT |
28 |
5.28% |
4.742 |
|
7.871 |
|
|
|
Biopsy only |
18 |
3.40% |
0.775 |
p<0.001 |
2.523 |
p<0.001 |
|
|
Biopsy + RT + CT |
115 |
21.70% |
5.059 |
|
6.882 |
|
|
|
Biopsy + CT |
60 |
11.32% |
0.698 |
|
2.651 |
|
|
|
Total |
530 |
100.0% |
6.267 |
|
12.360 |
|
|
Focusing on the type of CT received by patients, 29.8% were treated exclusively with temozolomide; 16.5% were treated with temozolomide + bevacizumab (bevacizumab in disease progression); 4.8% had bitherapy with temozolomide + fotemustine, administered at disease progression; and triple therapy (temozolomide + bevacizumab + fotemustine) was used in 10.1% of patients as sequential treatment in each progression. In total, 61.17% received treatment with temozolomide, either as monotherapy or in combination with another drug.
Regarding RT treatment, the most used fractionation in our series was 60 Gy (2 Gy fractions), which was administered to 339 patients (50.8 %), of which 65 (12.26 %) received a second RT treatment on the previously treated area. Two hundred twenty-one patients (33.2%) did not receive RT treatment during their oncological process. A total of 38 patients (7.16%) underwent a second surgical intervention.
Regarding the extension studies performed on patients, 448 patients (84.5%) underwent extension studies with computed tomography of the thorax, abdomen, and pelvis (CT TAP) or positron emission tomography-computed tomography (PET CT); none of these patients presented any sign of dissemination of the primary brain tumour outside the CNS. During follow-up of the oncological disease, complementary studies were performed to evaluate any possible dissemination of the primary disease; none of these studies showed disease outside the CNS. Neuroaxis dissemination was observed in 13 patients (2.45%) via MRI following changes in their neurological symptoms.
As for the cause of death, 96.6% of cases were due to disease progression at the CNS level, representing 512 of the 530 GBM patients. Mortality due to infections was the second cause in these patients, accounting for a total of 12 deaths (2.26%).
4.3. Progression-Free Survival (PFS)
Median PFS was 6.26 months (95% CI: 5.7-7.8). The univariate analysis showed that patients who received treatment with complete surgery + RT + CT had the longest relapse-free period, with a median of nine months. A statistically significant worst prognosis of < 1 month was found in patients treated with biopsy +/- CT (p < 0.05) (Figure 1). Regarding sex, no significant differences were found in terms of mean PFS (p = 0.458), being 5.9 months in women and 6.4 months in men.
4.4. Overall Survival (OS)
Median OS was 12.3 months (95% CI: 11.3-13.8) in patients diagnosed with GBM. The stratified study conducted on these GBM patients according to the type of treatment received revealed longer survival in patients who had received treatment with surgery + RT + CT (Figure 1). The five-year survival of GBM patients was 3.26%, while the one-year survival after diagnosis was 33.2%. If we differentiate patients by type of RT received, we found that standard treatment with 60 Gy was the most used and had the highest survival, 18.8 months; survival was less than 10 months with any other treatment scheme.
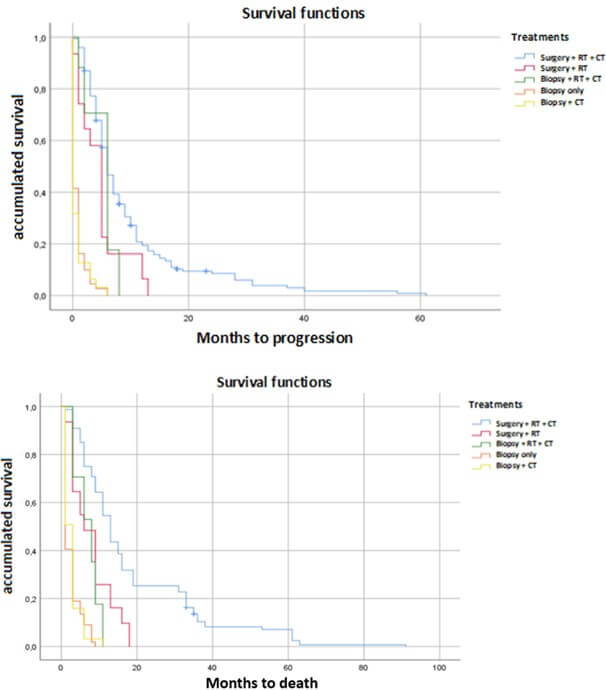
In terms of the type of CT, patients who received triple therapy (temozolomide + bevacizumab + fotemustine) presented the longest survival. In addition, when stratified for patients treated with or without bevacizumab, a statistically significant increased survival of 20.6 months (95% CI: 13.7-25.1) was observed in patients who received bevacizumab compared with 7.3 months (95% CI: 6.6-8.4) in those who did not (p < 0.05). Regarding sex, the univariate analysis demonstrated that women had a statistically significant worse survival (10 months) than men (13 months) (p < 0.05).
5. Anaplastic Astrocytoma Results
5.1. Patient Selection
In our search, we found a total of 1784 patients. However, there was a registry error for 727 patients, who had not been diagnosed with a primary brain tumour. Of these 727 patients, 523 were excluded as the brain tumour was metastatic from a primary tumour outside the CNS, and 205 because they did not have a definitive PA study (diagnosis was exclusively by imaging). Of the remaining 1,778 patients evaluated in the study (Table 1), 667 met the inclusion criteria for the proposed study. A total of 137 were diagnosed with AA.
If we examine AA diagnoses per year, to assess whether there has been an increase in incidence in recent years, we observe the following results:
2011: 9, 2012: 13, 2013: 10, 2014: 8, 2015: 14, 2016: 15, 2017: 13, 2018: 16, 2019: 19 and 2020: 20.
5.2. Clinical and Demographic Characteristics
The median age at diagnosis of the sample was 56 years (range: 25-88, SD: 14.59). The median age of death was 55.32 (range: 19-80, SD: 18.62). If we stratify AA into age groups of 18-30, 30-40, 40-60, and > 60 years, we observe that the population of AA patients is younger, with patients over 60 years of age representing less than 50% of cases (Table 2). In our cohort, there was a slightly higher percentage of men than women (53.5% and 46.5%, respectively). Regarding patients’ functional status measured by the ECOG scale at diagnosis, 55 patients (40.2%) had an ECOG of 0, 60 (43.8%) had an ECOG of 1, and 22 (16%) had an ECOG of 2 or higher. Regarding the treatment received by patients with AA, it is noteworthy that a large majority of patients were treated with surgery + RT + CT (73.72%). The second most common option was biopsy without any adjuvant treatment (Table 4). Regarding RT treatment, the most used fractionation in our series was 60 Gy (2 Gy fractions), which was administered to 88 patients (77.8%) of the 113 patients who received RT. The second most frequent RT dose option was 54 Gy (2 Gy fractions), administered to 20 patients (17.6%).
TABLE 4: Type of treatment received by patients with anaplastic astrocytoma and
survival results by type of treatment.
|
Treatments |
Total N |
|
Progression-free Survival (months) |
|
Overall Survival (months) |
|
|
Percentage |
|
|
|
|
||
|
Surgery + RT + CT |
101 |
73.72 |
34.255 |
|
50.287 |
|
|
Biopsy + RT + CT |
12 |
8.76 |
8.250 |
p<0.001 |
12.000 |
p<0.001 |
|
Biopsy only |
24 |
17.52 |
0.556 |
|
1.875 |
|
|
Overall |
137 |
100 |
25.068 |
|
38.453 |
|
5.3. Progression-Free Survival (PFS)
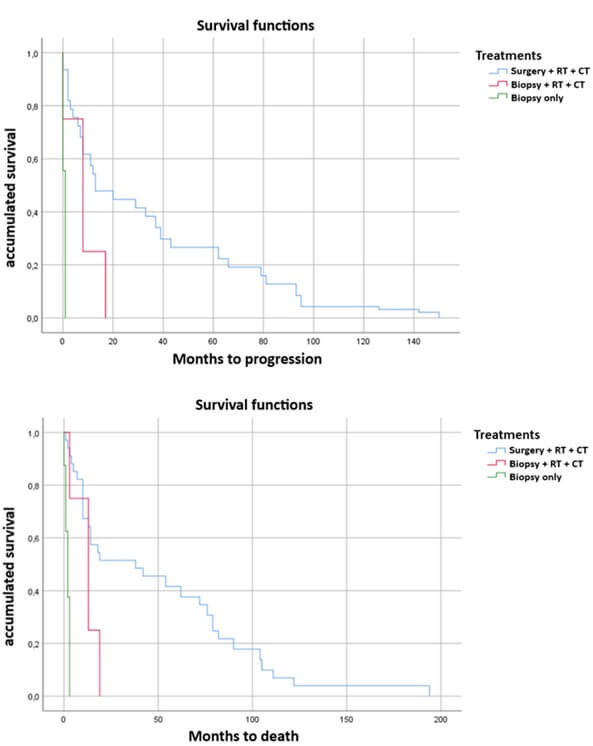
Median PFS was 25 months (95% CI: 20.7-31.9) in the group of AA patients. The multivariate analysis showed a significant positive association of PFS with the treatment option of complete surgery + RT + CT compared with patients who did not receive complete surgery, even if the RT + CT treatment was completed. Median time to relapse was 34.25 months (95% CI: 28.6-40.7) in patients who received surgery + RT + CT, and 8.25 months (95% CI: 7.1-8.9) in those patients who could not undergo surgery but did receive RT + CT (Figure 2). There were significant differences according to sex in this group, where being male afforded a protective factor with a median relapse-free survival of 33.85 months (95% CI: 27.67-41.5) compared with 17.62 months for women (p < 0.001).
5.4. Overall Survival (OS)
Median OS was 38.4 months (95% CI: 30.7-51.1) in AA patients. Prolonged survival was observed in patients treated with surgery + RT + CT, with a median of 50.28 months compared with 12 months in those patients who could not undergo surgery but did receive RT + CT. In patients with biopsy only, the median survival was two months (Figure 2). Regarding sex, male patients exhibited a statistically significant higher survival, with a median of 43.21 months (95% CI: 33.4-52.8) compared with 34.63 months (95% CI: 28.7-40.9) in female patients (p < 0.001). The five-year survival of AA patients was 29.92%, while the one-year survival was 75.92%.
6. Discussion
To date, some studies have evaluated the OS of these tumours; most reported results similar to those in our study, with survival times ranging from 8 to 29 months [15-18]. Historically, the median OS for GBM after the publication of the trial by Stupp et al. was established at 14.6 months [19]. However, the evolution of medicine in recent years, especially in the field of cancer treatment and diagnostic tests for this type of patient, has led to a slight improvement in the results [20, 21]. In our sample, a slight improvement in OS was observed, with a median survival of 18.6 months in patients treated with the same regimen as in the Stupp protocol.
On the other hand, OS exhibited a significant difference between the two groups, as in the current literature, where AA has greater survival for both PFS and OS (p < 0.001). When looking at sex, males had a protective factor in both subgroups. In the GBM subgroup, there was no significant difference in PFS between men and women (6.4 and 5.9 months, p = 0.458); however, for OS, being of the male gender had a protective factor in our sample, living a median of three months longer (p < 0.008). As for the AA group, the sex factor was also important, highlighting that being male was a protective factor since there was a significant difference both at the OS (43.21 months in male patients compared with 34.63 months in female patients) and PFS level (33.85 months for men and 17.62 months for women) (p < 0.001). Further studies, with a larger number of patients, are needed to explore this effect in this subset of tumours.
Overall, our data suggest greater OS in fully treated patients, with complete surgery being the feature of greatest benefit in terms of prolonging survival. As a rule, if the patient is in good general condition and there is progression, the cancer treatment is continued as there is increased survival in patients treated with different chemotherapy schemes [22]. Tumours harbouring methylation of the O6-methylguanine-DNA-methyltransferase (MGMT) gene are known to have a favourable response to temozolomide treatment [23], which could be a potential confounding factor for the differences observed among patients treated with CT.
Several studies found an association between survival outcomes and RT treatment, as well as improved clinical outcomes [24, 25]. The most common RT dose used in our study was 60 Gy, which is the habitual dose in these treatments [19]. Greater survival was found in patients receiving this dose compared with those who received lower doses, which could be related to the patient's condition or the location of the tumour. In addition, patients who underwent re-irradiation did not have improved survival. In these patients, the probability of extracranial disease is very low [26-28]; no disseminated disease was observed in our sample. The vast majority of patients had extension studies, only 15.5% did not have a brain tumour disease extension study, probably because the patients were considered palliative, given the extension of the disease at the brain level and the ill-fated prognosis. Functional status was better at diagnosis in the AA group, probably related to the younger age at diagnosis of this group.
When examining the incidence of GBM, it appears to be increasing. This finding is in line with several articles published to date [29-31]. The increase in incidence could be related to longer life expectancy due to a reduction in deaths from cardiovascular disease and other causes, although the reasons for this increase in incidence in recent years remain unclear. There is agreement that at least part of this increase is the result of improved diagnostic tests [1, 9]. However, improved diagnostic capacity cannot account for all this increase in the incidence of brain tumours. These data, along with evidence suggesting that the incidence may have been growing for many decades, leave open the possibility that environmental exposure may explain some of the rising incidence of brain tumours [1].
Within environmental exposure, ionising radiation is the only firmly established environmental risk factor for brain tumours. Cohort studies of atomic bomb and childhood cancer survivors have shown that cranial radiation is associated with an increased risk of various brain tumours, including meningiomas, gliomas, and nerve sheath tumours. In the case of non-ionising radiation, such as the use of mobile phones, a meta-analysis including data from 22 case-control series established that there was a slightly increased risk associated with mobile phone use; however, there were potential confounders [34]. Another conclusion of this meta-analysis was that the risk appeared after an induction period of 10 years or longer. Due to inconsistencies observed in studies and potential biases in case-control studies, in 2011, the WHO/IARC classified radiofrequency electromagnetic fields as possibly carcinogenic to humans (Group 2B, i.e., a causal association is considered credible, but when chance, bias, or confounding cannot be ruled out with reasonable confidence).
In addition, new causative agents such as radon have been observed in recent years. In the United States, a study was carried out suggesting a higher incidence of non-malignant brain tumours in regions with high exposure to particles and radon. These findings provide insight into the unexplained variation in tumour incidence, although future studies are needed to validate these findings in other populations [35].
7. Conclusion
In our experience, we have observed the aggressive potential of GBM, with very low survival rates (33% one-year survival), and a progressive increase in incidence in recent years. We have shown that surgery together with complete cancer treatment (CT+RT+QT) is the most important factor in OS. In terms of sex, male patients have longer survival, with a more marked difference in patients with AA. This group, who were younger, had a better survival, with an average of almost three years.
Limitations and Future Directions
This study has inherent limitations due to its retrospective nature and the relatively small size of the AA subgroup. There is also a risk that medical records may be inaccurate or inconsistent. The study was also subject to selection bias since data collection was carried out with a computerised system of medical records and some patients were collected at a time when the computerised medical history was not widely used, which could affect the results of the sample. On the other hand, a strength of this manuscript includes the large number of patients diagnosed with GBM.
Furthermore, this article may help us gain a better understanding of the incidence of this type of tumour and help pilot studies on organ donation in cancer patients with malignant primary brain tumours, such as that being developed at the Virgen de la Arrixaca University Clinical Hospital [36].
Conflicts of Interest
None.
REFERENCES
1. J Galceran, A Ameijide, M Carulla, et
al. “Cancer incidence in Spain, 2015.” Clin Transl Oncol, vol. 19, no.
7, pp. 799-825, 2017. View at: Publisher
Site
| PubMed
2. Sociedad española de oncologia médica.
Cifras del cancer en españa 2020. pp. 36, 2020.
3. Quinn T Ostrom, Nirav Patil, Gino
Cioffi, et al. “CBTRUS Statistical Report: Primary Brain and Other Central
Nervous System Tumors Diagnosed in the United States in 2013-2017.” Neuro
Oncol, vol. 22, no. 12 Suppl 2, pp. iv1-iv96, 2020. View at: Publisher Site | PubMed
4. Betsy A Kohler, Elizabeth Ward, Bridget
J McCarthy, et al. “Annual Report to the Nation on the Status of Cancer,
1975-2007, Featuring Tumors of the Brain and Other Nervous System.” J Natl
Cancer Inst, vol. 103, no. 9, pp. 714-736, 2011. View at: Publisher Site | PubMed
5. https://www.seom.org/seomcms/images/stories/recursos/Las_cifras_del_cancer_en_ Esp_2017.pdf.
6. https://seom.org/images/LAS_CIFRAS_DEL_CANCER_EN_ESPANA_2022.pdf.
7. https://seom.org/images/Las_cifras_del_Cancer_en_Espana_2023.pdf.
8. K Radhakrishnan, B Mokri, J E Parisi,
et al. “The trends in incidence of primary brain tumors in the population of
Rochester, Minnesota.” Ann Neurol, vol. 37, no. 1, pp. 67-73, 1995. View
at: Publisher
Site
| PubMed
9. A P Polednak “Interpretation of Secular
Increases in Incidence Rates for Primary Brain Cancer in Connecticut Adults,
1965-1988.” Neuroepidemiology, vol. 15, no. 1, pp. 51-56, 1996. View at:
Publisher
Site
| PubMed
10. Margaret Wrensch, Yuriko Minn, Terri
Chew, et al. “Epidemiology of primary brain tumors: Current concepts and review
of the literature.” Neuro Oncol, vol. 4, no. 4, pp. 278-299, 2002. View
at: Publisher
Site
| PubMed
11. F G Davis, S Freels, J Grutsch, et al.
“Survival rates in patients with primary malignant brain tumors stratified by
patient age and tumor histological type: an analysis based on Surveillance,
Epidemiology, and End Results (SEER) data, 1973-1991.” J Neurosurg, vol.
88, no. 1, pp. 1-10, 1998. View at: Publisher
Site
| PubMed
12. P Y Wen 1, H A Fine, P M Black, et al.
“High-grade astrocytomas.” Neurol Clin, vol. 13, no. 4, pp. 875-900, 1995. View
at: PubMed
13. Luis Enrique Contreras “EPIDEMIOLOGÍA
DE TUMORES CEREBRALES.” Revista Médica Clínica Las Condes, vol. 28, no.
3, pp. 332-338, 2017. View at: Publisher
Site
14. Sverre Helge Torp, Ole Solheim, Anne
Jarstein Skjulsvik “The WHO 2021 Classification of Central Nervous System
tumours: a practical update on what neurosurgeons need to know-a minireview.” Acta
Neurochir (Wien), vol. 164, no. 9, pp. 2453-2464, 2022. View at: Publisher
Site
| PubMed
15. Abudumijit Aibaidula, Aden Ka-Yin Chan,
Zhifeng Shi, et al. “Adult IDH wild-type lower-grade gliomas should be further
stratified.” Neuro Oncol, vol. 19, no. 10, pp. 1327-1337, 2017. View at:
Publisher
Site
| PubMed
16. Kosuke Aoki, Hideo Nakamura, Hiromichi
Suzuki, et al. “Prognostic relevance of genetic alterations in diffuse
lower-grade gliomas.” Neuro Oncol, vol. 20, no. 1, pp. 66-77, 2018. View
at: Publisher
Site
| PubMed
17. David E Reuss 1, Annekathrin Kratz,
Felix Sahm, et al. “Adult IDH wild type astro- cytomas biologically and
clinically resolve into other tumor entities.” Acta Neuropathol, vol.
130, no. 3, pp. 407-417, 2015. View at: Publisher
Site
| PubMed
18. Damian Stichel, Azadeh Ebrahimi, David
Reuss, et al. “Distribution of EGFR amplification, combined chromosome 7 gain
and chromosome 10 loss, and TERT promoter mutation in brain tumors and their
potential for the reclassification of IDHwt astrocytoma to glioblastoma.” Acta
Neuropathol, vol. 136, no. 5, pp. 793-803, 2018. View at: Publisher
Site
| PubMed
19. Roger Stupp, Warren P Mason, Martin J
van den Bent, et al. “Radiotherapy plus concomitant and adjuvant temozolo- mide
for glioblastoma.” N Engl J Med, vol. 352, no. 10, pp. 987-996, 2005.
View at: Publisher
Site
| PubMed
20. Wolfgang Wick, Thierry Gorlia, Martin
Bendszus, et al. “Lomustine and bevacizumab in progres- sive glioblastoma.” N
Engl J Med, vol. 377, no. 20, pp. 1954-1963, 2017. View at: Publisher Site | PubMed
21. Monika E Hegi, Annie-Claire Diserens,
Thierry Gorlia, et al. “MGMT gene silencing and benefit from temozolomide in
glioblastoma.” N Engl J Med, vol. 352, no. 10, pp. 997-1003, 2005. View
at: Publisher
Site
| PubMed
22. Ulrich Herrlinger, Theophilos Tzaridis,
Frederic Mack, et al. “Lomustine-temozolomide combination therapy versus
standard temozolomide therapy in patients with newly diagnosed glioblastoma
with methylated MGMT promoter (CeTeG/NOA-09): a randomised, open-label, phase 3
trial.” Lancet, vol. 393, no. 10172, pp. 678-688, 2019. View at: Publisher
Site
| PubMed
23. Andres Ramos-Fresnedo, Michael W
Pullen, Carlos Perez-Vega, et al. “The survival outcomes of molecular
glioblastoma IDH-wildtype: a multicenter study.” J Neurooncol, vol. 157,
no. 1, pp. 177-185, 2022. View at: Publisher
Site
| PubMed
24. Linda Chen, Hugo Guerrero-Cazares,
Xiaobu Ye, et al. “Increased subventricular zone radiation dose correlates with
survival in glioblastoma patients after gross total resection.” Int J Radiat
Oncol Biol Phys, vol. 86, no. 4, pp. 616-622, 2013. View at: Publisher
Site
| PubMed
25. Kyung Hwan Kim, Jihwan Yoo, Nalee Kim,
et al. “Efficacy of whole-ventricular radiotherapy in patients undergoing
maximal tumor resection for glioblastomas involving the ventricle.” Front
Oncol, vol. 11, pp. 736482, 2021. View at: Publisher Site | PubMed
26. Manish J Gandhi, D Michael Strong
“Donor derived malignancy following transplantation: a review.” Cell Tissue
Bank, vol. 8, no.4, pp. 267-286, 2007. View at: Publisher
Site
| PubMed
27. P Beauchesne, C Soler, J F “Mosnier
Diffuse vertebral body metastasis from a glioblastoma multiforme: a
technetium-99m Sestamibi single-photon emisión computerized tomography study:
Case report.” J Neurosurg, vol. 93, no. 5, pp. 887-890, 2000. View at: Publisher
Site
| PubMed
28. A Didelot, L Taillandier, Y Grignon, et
al. “Concomitant bone marrow metastasis of a glioblastoma multiforme revealed
at the diagnosis.” Acta Neurochir, vol. 148, no. 9, pp. 997-1000, 2006.
View at: Publisher
Site
| PubMed
29. Therese A Dolecek, Jennifer M Propp,
Nancy E Stroup, et al. “CBTRUS statistical report: primary brain and central
nervous system tumors diagnosed in the United States in 2005-2009.” Neuro
Oncol, vol. 14 Suppl 5, no. Suppl 5, pp. v1-v49, 2012. View at: Publisher Site | PubMed
30. Sundeep Deorah, Charles F Lynch, Zita A
Sibenaller, et al. “Trends in brain cancer incidence and survival in the United
States: surveillance, epidemiology, and end results program, 1973 to 2001.” Neurosurg
Focus, vol. 20, no. 4, pp. E1, 2006. View at: Publisher Site | PubMed
31. Miikka Korja, Rahul Raj, Karri Seppä,
et al. “Glioblastoma survival is improving despite increasing incidence rates:
a nationwide study between 2000 and 2013 in Finland.” Neuro Oncol, vol.
21, no. 3, pp. 370-379, 2019. View at: Publisher Site | PubMed
32. A P Polednak “Interpretation of Secular
Increases in Incidence Rates for Primary Brain Cancer in Connecticut Adults,
1965-1988.” Neuroepidemiology, vol. 15, no. 1, pp. 51-56, 1996. View at:
Publisher
Site
| PubMed
33. J Galceran, A Ameijide, M Carulla, et
al. “Cancer incidence in Spain, 2015.” Clin Transl Oncol, vol. 19, no.
7, pp. 799-825, 2017. View at: Publisher
Site
| PubMed
34. Seung-Kwon Myung, Woong Ju, Diana D
McDonnell, et al. “Mobile phone use and risk of tumors: a meta-analysis.” J
Clin Oncol, vol. 27, no. 33, pp. 5565-5572, 2009. View at: Publisher Site | PubMed
35. Joshua D Palmer, Rahul N Prasad, Gino
Cioffi, et al. “Exposure to radon and heavy particulate pollution and incidence
of brain tumors.” Neuro Oncol, vol. 25, no. 2, pp. 407-417, 2023. View
at: Publisher
Site
| PubMed
36. Juan Antonio Encarnación Navarro “Donación de Órganos en Pacientes con Tumor Maligno Primario Cerebral: Posibles Donantes en la Región de Murcia. Análisis de los Registros Internacionales y Nacionales.” Universidad de Murcia.
