Received: Tue 15, Apr 2025
Accepted: Tue 06, May 2025
Abstract
Background: Currently, the primary treatment for ulnar neuropathy at the elbow is open in-situ decompression surgery. The effectiveness of ultrasound localization therapy, especially small-incision surgery using ultrasound combined with SWE, remains unclear.
Objectives: To evaluate the effect of small-incision ulnar nerve release in treating ulnar neuropathy at the elbow by ultrasound combined with shear wave elastography (SWE).
Methods: A retrospective analysis of 98 patients treated in our hospital for ulnar neuropathy at the elbow was conducted from June 2023 to March 2025. According to the treatment style, these patients were divided into a traditional open in-situ decompression surgery group (n = 51) and an ultrasound combined with SWE small-incision surgery group (n = 47). The maximum proximal Cross-sectional Area (CSA), sensory conduction velocity (SCV), motor conduction velocity (MCV), modified Bishop score, Quick-DASH score, and visual analogue scale (VAS) score were compared between the two groups. Additionally, the amount of intraoperative blood loss, operation duration, hospital stay, patients' satisfaction with postoperative incision aesthetics, the postoperative complications of different operation methods, and the degree of damage to the medial cutaneous nerve of the forearm were studied.
Results: All patients underwent surgical treatment, with preoperative ultrasonography confirming no ulnar nerve subluxation. No statistically significant differences in age, preoperative SCV, MCV, CSA, Quick-DASH score, or VAS score were found between the two groups. The postoperative and the last follow-up SCV, MCV, and CSA were similar in the two groups. In addition, the improved Bishop score, Quick-DASH score, VAS score, postoperative hematoma rate, elbow stiffness rate, and postoperative protection of medial cutaneous nerve of the forearm in the ultrasound combined with SWE surgery group were better than those in the traditional open surgery group. What’s more, compared with the traditional open surgery group, the ultrasound combined with SWE surgery reduced the amount of intraoperative blood loss, shortened the operation duration and hospital stay, and the patients were more satisfied with the scar of the incision.
Conclusion: Preoperative ultrasound combined with SWE can accurately and comprehensively evaluate the ulnar nerve compression site so as to provide effective guidance for the precise and minimally invasive treatment of ulnar neuropathy at the elbow.
Keywords
Ulnar neuropathy, ultrasound, SWE, in situ decompression, small incision, medial cutaneous nerve of forearm
1. Introduction
Ulnar neuropathy is the second most common upper limb compression neuropathy after carpal tunnel syndrome [1]. The main signs and symptoms of ulnar neuropathy at the elbow are as follows [2]: numbness and pain in the ulnar innervation area (especially in the ring finger and fifth digit), fine motor disorders of the hand, burning sensation in the forearm and hand, and advanced sensory loss. These symptoms can worsen with elbow bending during sleep at night. To diagnose it, electromyography (EMG) results must meet at least two of the following four criteria [3]. i) Nerve conduction velocity (NCV) from upper elbow to lower elbow (AE to BE) is less than 50 m/s. ii) NCV from AE to BE is slower than NCV from BE to wrist by more than 10 m/s. iii) Compound motor action potential (CMAP) decreased by more than 20% from BE to AE. iv) Changes of CMAP waveform between AE and BE. However, Cristiani-Winer et al. [4] found that EMG is not very valuable in diagnosing ulnar neuropathy at the elbow, while ultrasound offers greater specificity, requires less time, is more cost-effective, and is non-invasive, making it more applicable in clinical practice. The CSA of the ulnar nerve measured by ultrasound imaging ≥ 10mm2 can be used as the gold standard for diagnosing ulnar neuropathy [5, 6]. Although the conservative approach can reduce pain and delay the progression of ulnar nerve compression to some extent, surgical decompression is still the main treatment for moderate to severe ulnar neuropathy at the elbow [7]. At present, the mainstream surgical methods include open decompression release and endoscopic release. Although traditional open surgery is thorough in decompression, its large incision is more likely to cause damage to peripheral nerve vessels. Moreover, studies have indicated that minimally invasive techniques, like endoscopic release, have higher rates of postoperative complications, such as hematoma and reoperation, compared to open decompression [8]. However, these methods result in significantly lower chronic pain levels after surgery [7]. Therefore, less trauma, better outcomes, and a lower risk of postoperative complications are what we are looking for.
Ultrasound is crucial for diagnosing ulnar neuropathy, and ultrasound-assisted ulnar nerve decompression therapy offers several advantages over other treatments. For example, compared with electrodiagnosis and magnetic resonance imaging, ultrasound is better tolerated, less costly, and less time-consuming, making ultrasound-assisted ulnar nerve release therapy feasible [9]. Shear wave elastography (SWE), a new ultrasound imaging technology, can quantitatively evaluate the hardness of peripheral nerves, thereby improving the accuracy of ultrasonic diagnosis and localization of peripheral neuropathy [10, 11]. Recent studies indicate that while in situ decompression using traditional ultrasonic guidance minimizes surgical trauma, it lacks the accuracy needed to diagnose early lesion entrapments. Additionally, it has limited effectiveness in evaluating ulnar neuropathy, which can lead to unsatisfactory postoperative outcomes [12]. Therefore, the purpose of this study was to retrospectively analyze the patients with ulnar neuropathy at the elbow who received surgical treatment in our hospital, and compare the efficacy of traditional open in-situ decompression, and small-incision modified in-situ decompression with ultrasound combined with SWE, to evaluate the feasibility and application prospect of ultrasound-combined SWE precision positioning in the treatment of ulnar neuropathy at the elbow.
2. Methods
2.1. General Information and Grouping Methods
Ninety-eight patients with ulnar neuropathy at the elbow who underwent surgical treatment in our hospital from June 2023 to March 2025 were retrospectively analyzed. The inclusion criteria were as follows: i) Age > 18 years. ii) Obvious symptoms of ulnar nerve compression. iii) Preoperative EMG showed that the ulnar nerve motor conduction velocity (MCV) of the elbow was < 50 m/s. iv) Preoperative ultrasonography showed ulnar nerve compression around the cubital canal, with CSA ≥ 10mm2. Exclusion criteria: i) Postoperative revision of previous ulnar neuropathy at the elbow. ii) Ulnar nerve compression caused by neuro-neoplasms, rheumatoid arthritis, or tuberculosis of the elbow. iii) Simultaneous carpal tunnel syndrome, thoracic outlet syndrome, and other neurological disorders. iv) Incomplete clinical data.
A total of 98 cases were included, including 53 males and 45 females. All patients underwent successful decompression surgery. According to different surgical methods, they were divided into a traditional open in situ decompression surgery group (n = 51) and an ultrasound combined with SWE small-incision surgery group (n = 47). This study was approved by the hospital's Clinical Research Ethics Committee (Approval No.2023-KY-233-01) and meets the ethical standards of the Declaration of Helsinki. Informed consent was obtained from all patients participating in the study.
2.2. Ultrasound and Surgical Procedure
All operations were performed under standard procedures (arm overhang and tourniquet inflation). The shoulders were in the abduction and rotation position, and the elbows were in the 90° flexion position. Routine disinfection and paving.
2.2.1. Traditional Open in situ Decompression
A 7-10 cm arc incision was made with the posterior margin of the internal epicondyle of the humerus as the center, the skin and subcutaneous tissue were separated, and the ulnar nerve was carefully found and exposed by walking along the direction between the internal epicondyle of the humerus and the olecranon. Then, the medial muscular septum, Struthers' arcade, Osborne ligament, and the tendinous membrane of the ulnar flexor carpi ulnaris were released successively from the proximal end to the distal end. Attention was paid to protecting nerve branches and accompanying blood vessels. After the operation, the incision was fully rinsed with iodophor and normal saline, and then sutured layer by layer, covered with gauze, and fixed with a bandage.
2.2.2. In situ Decompression under Ultrasound Combined with SWE
GE LOGIQ E11 color doppler ultrasonic diagnostic instrument was used, with a 6- 15 MHz linear array probe, and MSK mode was selected. The patient was placed in a supine position, with the upper limbs relaxed naturally, abduction and external rotation about 60° ~ 75°, palms facing upward, and fingers naturally flexed. Continuous transverse and longitudinal scanning were performed on the ulnar nerve 5 cm above and below the cubital canal to observe the shape of the ulnar nerve, internal echo, and the relationship between the ulnar nerve and surrounding tissues, and to determine whether there is an incisal sign, that is, the compression position. The maximum cross-sectional area (CSA) of the ulnar nerve was measured by describing the hyperechoic edge inside the ulnar nerve by enlargement. Then, the probe was rotated 90° along the long axis of the ulnar nerve to observe the location of the narrowest and most swollen ulnar nerve. Stabilized the probe, applied a large amount of coupling agent, lightly touched the skin, avoided pressurization, started SWE imaging mode, adjusted the depth to 2.5 cm, and set the range to 0 ~ 15 cm/s. Widened the region of interest (ROI) and selected the yellow or blue area when the image was stabilized. Images with color filling of more than 50% of the sample frame area and well-filled images were measured. The shear wave velocity (V) and the mean elastic modulus of the ulnar nerve in the ROI at the proximal end of the compression were measured, and then the elasticity index (EI) was calculated. Detection box ROI from red to blue represents tissue texture from soft to hard. More than 5 measured values were obtained. Elastic data was reliable when IQR/Median < 30%, and the mean was taken as the test result and recorded.
Diagnostic criteria of ultrasound combined with SWE: ① CSA≥10mm2 under conventional ultrasound. ② In SWE mode, yellow or blue areas are predominant. ③ The ulnar nerve stiffness was ≥ 61 kPa, the cubital tunnel (CT) to distal arm (DA) stiffness ratio was ≥ 1.68, and the CT to mid arm (MA) stiffness ratio was ≥ 1.75 [13]. The positions that met the above conditions were defined as the compression sites. All the above parameters were measured by the same musculoskeletal sonographer with extensive experience in elastic manipulation.
Took the compression position as the center and marked the 2-3 cm area above and below the center as the incision range. Cut the skin and subcutaneous tissue, exposed the entrapment ulnar nerve, released the tissue and ligament of the ulnar nerve layer by layer, and paid attention to protecting the surrounding blood vessels and other nerves. After the operation, the incision was rinsed with iodophor and normal saline, and then sutured layer by layer, covered with gauze, and fixed by applying. Both groups were fixed in the flexion position with plaster, given neurotrophic drugs, immobilized in the affected limb, placed on bed rest, and had sutures removed 14 days after surgery.
2.3. Clinical Efficacy Analysis and Follow-Up Plan
Operation time, intraoperative blood loss, postoperative pain relief, postoperative clinical symptom improvement, ulnar nerve compression relief, changes in SCV and MCV of the ulnar nerve, and aesthetic satisfaction of patients with surgical incision scars were recorded and compared in the two groups. The visual analog scale (VAS) [14] was used to evaluate the pain relief of patients in the two groups. Improved Bishop score [15, 16] and Quick-DASH score [17, 18] were used to evaluate the improvement of postoperative clinical symptoms in the two groups. The ulnar nerve compression and the degree of relief after the operation were evaluated by ultrasound, and the CSA of the proximal ulnar nerve compression was recorded. The changes in the SCV and MCV of the ulnar nerve before and after the operation were evaluated by electromyography. Aesthetic satisfaction scores (satisfaction = 3 points, general = 2 points, dissatisfaction = 1 point) were used to evaluate the satisfaction of the two groups of patients with postoperative scars. The patients discharged after the operation were followed up by telephone and imaging examination in the hospital to evaluate the long-term efficacy and postoperative complications.
2.4. Statistical Methods
SPSS 25.0 was used for statistical analysis. The measurement data were tested for normal distribution and represented by mean ± standard deviation. The age, SCV, MCV, CSA of proximal ulnar nerve compression of the elbow, modified Bishop score, Quick-DASH score, and VAS score of patients in different groups were compared by the T-test. The ulnar nerve SCV, MCV, CSA, Quick-DASH score, and VAS score of the same group of patients before and after the operation and at the last follow-up were compared by Repeated Measures Analysis of Variance (RM-ANOVA), and P < 0.05 was considered statistically significant. Counting data such as postoperative hematoma, incision infection, postoperative numbness, elbow stiffness, and postoperative medial cutaneous nerve damage of the forearm were expressed by frequency and percentage, and the Chi-square test or Fisher's exact test was used for comparison. When P < 0.05, the difference between the two groups was considered statistically significant.
3. Results
A total of 98 patients with ulnar neuropathy of the elbow were included in this study, all of whom underwent successful surgery. There were no statistical differences in gender, age, preoperative SCV, MCV, and CSA between the two groups. The post-operation and last follow-up SCV, MCV, and CSA of the small incision group under ultrasound combined with SWE were similar to those of the traditional surgery group. This suggests that small incision surgery under ultrasound combined with SWE can achieve a similar degree of ulnar nerve relaxation as open surgery (Table 1). The modified Bishop score, Quick-DASH score, and VAS score of the ultrasound combined with SWE small incision group were better than those of the traditional open surgery group at postoperative and last follow-up. In addition, the small incision group reduced the amount of intraoperative blood loss and shortened the operation time and hospital stay. Moreover, patients had higher satisfaction with postoperative incision scars (Table 2). In terms of postoperative complications, small-incision of ultrasound combined with SWE significantly reduced the incidence of postoperative hematoma and elbow joint stiffness and had a better protective effect on the medial cutaneous nerve of the forearm (Table 3). A typical case is shown in (Figure 1). Preoperative ultrasound and SWE images of the case are shown in (Figure 2).
TABLE 1: Basic
data and imaging indicators of the three groups of patients (mean ± SD).
|
|
Traditional open surgery group (n=51) |
Ultrasound combined with SWE (n=47) |
|
Sex (male/female, n) |
28/23 |
25/22 |
|
Age, years |
47.82±12.32 |
45.30±11.23 |
|
SCV, m/s Pre-operation Post-operation Last follow-up MCV, m/s |
40.44±2.48 44.77±2.08① 50.47±1.27① |
39.85±1.61 44.07±1.80① 50.11±1.60① |
|
Pre-operation |
32.20±1.21 |
32.32±1.18 |
|
41.55±1.49① |
41.42±1.40① |
|
|
Last follow-up |
52.55±1.41① |
52.10±1.34① |
|
CSA, mm2 |
|
|
|
Pre-operation |
13.03±2.28 |
13.31±2.50 |
|
Post-operation |
11.39±1.82① |
11.51±1.81① |
|
Last follow-up |
8.99±0.93① |
9.15±0.77① |
Note: ① Compared with
pre-operation, P < 0.05; ② Compared
with the traditional open surgery group, P < 0.05
SCV: Sensory Conduction Velocity; MCV: Motor Conduction Velocity; CSA: Cross-sectional Area.
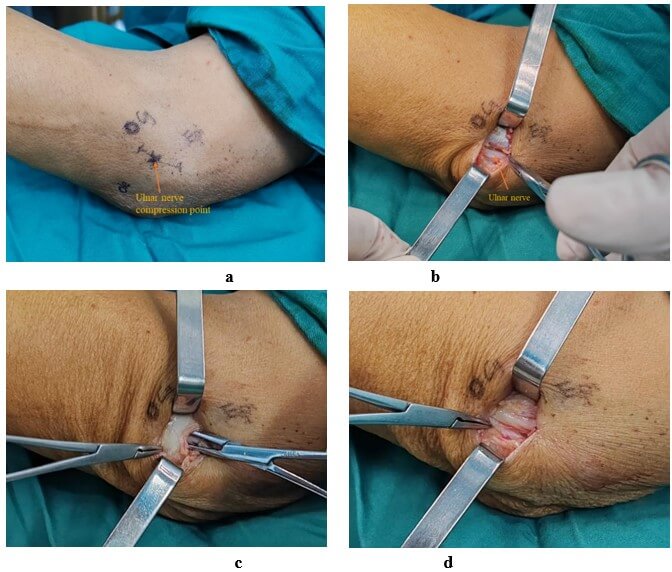
TABLE 2:
Indicators related to surgery (mean ± SD).
|
|
Traditional open surgery group (n=51) |
Ultrasound combined with SWE (n=47) |
|
Improved Bishop score, points |
6.43±0.50 |
7.55±1.16② |
|
Quick-DASH score, points Pre-operation Post-operation Last follow-up
VAS score, points Pre-operation Post-operation Last follow-up
Operation time, minutes Intraoperative blood loss, ml Postoperative hospital stay, hours ASS, points |
57.29±1.65 34.67±2.19① 25.10±1.84①
6.76±0.82 4.37±0.56① 2.27±0.67①
33.06±5.27 26.02±4.89 47.71±4.88 1.55±0.50 |
57.02±1.54 31.45±2.14①② 21.55±2.45①②
6.96±0.81 3.04±0.69①② 1.49±0.55①②
23.15±3.51② 10.81±2.84② 17.98±3.23② 2.60±0.50② |
Note: ① Compared with
pre-operation, P < 0.05; ② Compared
with the traditional open surgery group, P < 0.05
VAS: Visual Analogue Scale; ASS: Aesthetic Satisfaction Score.
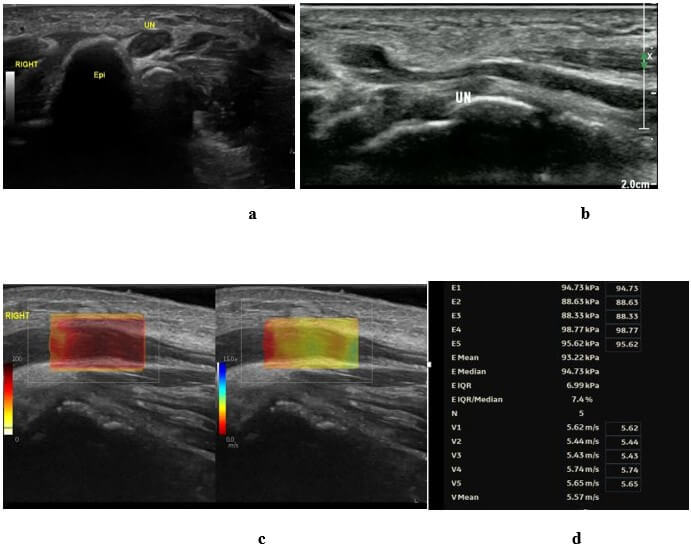
UN: Ulnar Nerve; Epi: Medial Condyle of Humerus.
TABLE 3:
Postoperative complications.
|
|
Traditional open surgery group (n=51) |
Ultrasound combined with SWE (n=47) |
|
Postoperative hematoma (n, %) |
6, 11.8% |
0, 0%* |
|
Incision infection (n, %) Postoperative numbness (n, %) Elbow stiffness (n, %) Postoperative cutaneous nerve lesion of medial forearm (n, %) |
3, 5.9% 5, 9.8% 7, 13.7% 8, 15.7%
|
1, 2.1% 2, 4.3% 0, 0%* 1, 2.1%*
|
* Compared with traditional open surgery group, P < 0.05.
4. Discussion
Ulnar neuropathy at the elbow is the second most common disease of upper limb peripheral nerve entrapment, with an annual incidence rate of 0.44‰ [19]. There are five common sites where the ulnar nerve may become compressed around the elbow: the medial interosseous interval, the Struthers arcade, the exostosis of the medial epicondyle of the humerus, the ligament of Osborne, and the tendinous membrane of the ulnar flexor carpi ulnaris. Among these, the tendinous membrane of the ulnar flexor carpi ulnaris is the most frequent point of compression [20]. Compression of the ulnar nerve leads to chronic ischemia and hypoxia, increased vascular permeability leading to nerve edema, and eventually a vicious cycle of ischemia, hypoxia, and edema, as well as nerve fiber tissue hyperplasia and Waller's degeneration [21]. The principle of treatment is to relieve these compressions to terminate the vicious cycle of nerve edema. Conventional open in situ decompression surgery involves sequential release of the medial interosseous interval, Struthers' arcade, Osborne's ligament, and ulnar flexor carpi radialis tendon membrane from proximal to distal along the course of the ulnar nerve between the medial epicondyle of the humerus and the falciform humerus [20]. Although open surgery can effectively relieve existing and potential sites of ulnar nerve compression, the large incision increases the risk of injuring surrounding blood vessels and nerves, and patients often express dissatisfaction with the appearance of the surgical scars [22]. Ultrasound-assisted localization of in situ decompression surgery cannot only accurately and comprehensively locate the point of compression of the ulnar nerve at the elbow but also has great significance for application in clinical work due to its minimally invasive nature and the aesthetics of surgical scarring [9, 12].
In terms of cost-effectiveness, ultrasound demonstrates significant advantages. Ho et al. [23] compared various diagnostic modalities for ulnar neuropathy at the elbow, demonstrating that MR neurography and diffusion tensor imaging achieve high localization accuracy. However, MR examinations incur substantial costs and lack real-time assessment capability. Neuro-electrophysiology can detect nerve conduction velocity and determine nerve function, and it is the gold standard for diagnosing ulnar neuropathy. However, it is an invasive examination and cannot visualize the nerve course and surrounding anatomical structures, with a false negative rate of 20% [24]. Both MR and neuro-electrophysiology are time-consuming and costly, limiting their utility as screening tools in clinical practice. In contrast, two-dimensional ultrasound offers non-invasive, rapid, and dynamic real-time evaluation of the entire nerve, clearly delineating ulnar nerve morphological characteristics and adjacent anatomical structures [25, 26]. When combined with SWE, which serves as an adjunct to conventional grayscale imaging, this modality enables early detection of neuropathies at a lower cost with high diagnostic yield. SWE quantitatively assesses tissue stiffness by measuring shear wave propagation velocity and has been widely applied in differentiating benign/malignant lesions and evaluating acute/chronic injuries across multiple systems, including hepatic, breast, thyroid, intestinal, and musculoskeletal tissues [27, 28]. By providing objective numerical values to quantify tissue elasticity and localize regional stiffness heterogeneity, SWE integrates seamlessly with standard ultrasound systems. This combined approach maintains operational simplicity while demonstrating high stability and reproducibility [10, 29, 30], facilitating early lesion detection and guiding clinical management/therapeutic monitoring. The established clinical and research applications of ultrasound combined with SWE confirm its role as an essential tool in modern ultrasonography, providing both theoretical and practical foundations for our investigation. However, most of the current applications of ultrasound in ulnar neuropathy at the elbow are limited to diagnosis, and there is no precedent for auxiliary treatment, especially small-incision decompression with precise localization. Our study, on the other hand, precisely bridges this gap.
The value of ultrasound in the diagnosis and management of ulnar neuropathy at the elbow has been shown to be simpler and more economical than MRI, and ultrasound can be used as an early indicator of patients with negative EMG results [31-33]. The CSA of the ulnar nerve measured by ultrasound ≥ 10mm2 can be used as a standard for diagnosing ulnar neuropathy at the elbow [5, 34]. Therefore, in our study, the CSA of proximal ulnar nerve compression at the elbow was used as one of the indices to evaluate the postoperative efficacy. We found no statistical difference in the CSA of the ulnar nerve between the small incision group under ultrasound combined with SWE and the traditional open surgery group, both post-surgery and at the last follow-up. This suggests that small incision decompression using ultrasound combined with SWE precise positioning can effectively release the compressed ulnar nerve, similar to open surgery. Additionally, the small incision surgery group benefited from more pronounced postoperative pain relief. This improvement may be attributed to milder nerve pulling, less tissue destruction, and a milder degree of tissue edema. Moreover, SWE, as an emerging non-invasive imaging technique for evaluating peripheral neuropathy, has promising application value in evaluating tissue hardness [35]. Ulnar neuropathy at the elbow, due to nerve compression, the pressure in the ulnar nerve canal increases, resulting in ischemia, edema, and inflammation, and eventually leads to intraneural space and synovial fibrosis, thus increasing nerve hardness, which is also the theoretical basis for SWE to detect cubital tunnel syndrome [10, 36]. PALUCH et al. [37] found that the nerve hardness of patients with ulnar neuropathy at the elbow was significantly higher than that of healthy people, and SWE played an important role in the diagnosis of ulnar neuropathy. However, there are limited reports on the efficacy of modified in situ decompression surgery using ultrasound combined with SWE precision positioning, and the risk/benefit ratio compared with traditional open surgery is unclear. In our study, patients with ulnar neuropathy at the elbow who received surgical treatment in our hospital were divided into a traditional open in situ decompression surgery group and an ultrasound combined with SWE small-incision surgery group according to different operation methods. The results showed that compared to traditional open in situ decompression surgery, small incision ulnar nerve release using ultrasound combined with SWE can significantly shorten operation time, reduce intraoperative blood loss, and lessen hospital stays. Moreover, the smaller incision improves patient satisfaction with scar aesthetics and minimizes the risk of complications such as postoperative hematoma, numbness, elbow stiffness, and nerve damage, promoting better recovery.
However, for traditional open surgery, the study of CELLI et al. [38] showed that ulnar nerve transposition could release all potential nerve compression points and establish a new path for the ulnar nerve. When the release was complete in the entire ulnar nerve path, it was recommended to perform ulnar nerve transposition if the epicondylar sulci were found to be too shallow or unstable. However, due to the small incision of the modified operation under ultrasonic precise positioning, it is difficult to continue the ulnar nerve transposition after in situ release. Therefore, in our study, patients requiring ulnar nerve transposition were excluded, thus avoiding the error caused by sampling bias. In addition, Wang et al. [22] found in their study that compared with traditional open in situ decompression, there was no statistical difference in the rate of reoperation between the two methods. On the contrary, open in situ decompression with large incisions resulted in more serious postoperative scar formation due to greater damage to surrounding tissues, and the possibility of re-compression of the ulnar nerve increased. Therefore, we can speculate that as long as all potential ulnar nerve compression sites are accurately positioned and completely released under ultrasound guidance during the operation, minimally invasive surgery with small incisions will not increase the risk of postoperative recurrence. The small incision in situ decompression using ultrasound combined with SWE precise positioning should be optimistic in terms of long-term efficacy. Of course, this also requires us to conduct more long-term follow-ups of patients in the later period to verify our conjecture.
However, there are some limitations in this study. First of all, this study is a single-center retrospective study with a small number of cases. Although certain clinical results have been achieved, multicenter prospective randomized controlled studies with a larger sample size are still needed to further verify its clinical efficacy. Second, the follow-up time of this study was short, and no further study was conducted on the long-term prognosis and postoperative revision rate. Longer follow-up is therefore needed to further confirm our conclusions. In addition, this study did not analyze the postoperative efficacy of patients with different degrees of ulnar nerve compression. In the future, the authors aim to grade the degree of ulnar nerve compression according to the maximum proximal CSA to determine the long-term efficacy of modified ulnar nerve release under ultrasound-precise localization for people with different degrees of compression.
5. Conclusion
Small incision in situ ulnar nerve release using ultrasound combined with SWE precise positioning has a definite effect on patients with ulnar neuropathy at the elbow. Compared with traditional open in-situ decompression, it can obtain a shorter recovery time and reduce the risk of postoperative complications, and patients have a higher satisfaction with postoperative scar aesthetics, which can be used for further clinical promotion.
Funding
This work was supported by the National Natural Science Foundation of China (No 82202823).
Data Availability
The datasets generated and analyzed during this study are available from the corresponding author upon reasonable request.
Author Contributions
KY, YH and TW designed the study. YH, YF, and XW were responsible for collecting, analyzing, and interpreting the data and writing the manuscript. JZ, XL, and KY identified the case and performed the surgery. MM was responsible for revising the manuscript. All authors read and approved the final manuscript.
Acknowledgments
The authors thank all patients and their families who participated in this study.
Ethics Approval and Consent to Participate
This study was approved by the hospital's Clinical Research Ethics Committee (Approval No.2023-KY-233-01) and meets the ethical standards of the Declaration of Helsinki. Informed consent was obtained from all patients participating in the study.
Consent for Publication
Not applicable.
Clinical Trial Number
Not applicable.
Competing Interests
None.
REFERENCES
[1] Munish Chauhan, Prashanth Anand, Joe M. Das “Cubital
Tunnel Syndrome.” StatPearls [Internet]. Treasure Island (FL): StatPearls
Publishing, 2024. View at: PubMed
[2] Saurabh Shelke, Ratnakar Ambade, Aditi Shelke “From
Conservative Measures to Surgical Interventions, Treatment Approaches for
Cubital Tunnel Syndrome: A Comprehensive Review.” Cureus, vol. 15, no.
12, pp. e51262, 2023. View at: Publisher Site | PubMed
[3] Joost T P Kortlever, Berdien Brandsema, Gertraud
Gradl-Dietsch, et al. “Electrodiagnostic test results in people with a working
diagnosis of cubital tunnel syndrome.” J Orthop, vol. 36, pp. 114-119,
2023. View at: Publisher Site | PubMed
[4] M Cristiani-Winer, C Allende-Nores, G L Aguirre, et
al. “Sensitivity and specificity of three complementary methods in the
diagnosis of carpal tunnel syndrome.” Acta Ortop Mex, vol. 34, no. 1,
pp. 31-37, 2020. View at: PubMed
[5] Ke-Vin Chang, Wei-Ting Wu, Der-Sheng Han, et al.
“Ulnar Nerve Cross-Sectional Area for the Diagnosis of Cubital Tunnel Syndrome:
A Meta-Analysis of Ultrasonographic Measurements.” Arch Phys Med Rehabil,
vol. 99, no. 4, pp. 743-757, 2018. View at: Publisher Site | PubMed
[6] Katherine McGurk, Joseph Anthony Tracey, Dane N Daley,
et al. “Diagnostic Considerations in Compressive Neuropathies.” J Hand Surg
Glob Online, vol. 5, no. 4, pp. 525-535, 2022. View at: Publisher Site | PubMed
[7] Eslam Abourisha, Ananth S Srinivasan, Ahmed Barakat,
et al. “Surgical management of cubital tunnel syndrome: A systematic review and
meta-analysis of randomised trials.” J Orthop, vol. 53, pp. 41-48, 2024.
View at: Publisher Site | PubMed
[8] Patrick J Buchanan, Lee O Chieng, Zachary S Hubbard,
et al. “Endoscopic versus Open In Situ Cubital Tunnel Release: A Systematic
Review of the Literature and Meta-Analysis of 655 Patients.” Plast Reconstr
Surg, vol. 141, no. 3, pp. 679-684, 2018. View at: Publisher Site | PubMed
[9] Naina S Limbekar, Maximillian C Soong, Michal M
Vytopil, et al. “High-Resolution Ultrasound in the Diagnosis and Surgical
Management of Ulnar Neuropathy at the Elbow.” Orthopedics, vol. 44, no.
5, pp. 285-288, 2021. View at: Publisher Site | PubMed
[10]
Sujin Kim,
Guen Young Lee “Evaluation of the ulnar nerve with shear-wave elastography: a
potential sonographic method for the diagnosis of ulnar neuropathy.” Ultrasonography,
vol. 40, no. 3, pp. 349-356, 2021. View at: Publisher Site | PubMed
[11]
Lihua Liu,
Tiezheng Wang, Hengtao Qi, et al. “Multimodal ultrasound evaluation of
asymptomatic ulnar nerve dislocation at the cubital tunnel.” Quant Imaging
Med Surg, vol. 14, no. 1, pp. 633-639, 2024. View at: Publisher Site | PubMed
[12]
Jin-Mei Gao,
Yu Yuan, Ke-Tong Gong, et al. “Ultrasound-Assisted Precise In Situ
Decompression for Cubital Tunnel Syndrome.” Orthop Surg, vol. 13, no. 3,
pp. 840-846, 2021. View at: Publisher Site | PubMed
[13]
Łukasz Paluch,
Bartłomiej Noszczyk, Żaneta Nitek, et al. “Shear-wave elastography: a new potential method to diagnose ulnar
neuropathy at the elbow.” Eur Radiol, vol. 28, no. 12, pp. 4932-4939, 2018. View at: Publisher Site | PubMed
[14]
Mark P Jensen,
Connie Chen, Andrew M Brugger “Interpretation of visual analog scale ratings
and change scores: a reanalysis of two clinical trials of postoperative pain.” J
Pain, vol. 4, no. 7, pp. 407-414, 2003. View at: Publisher Site | PubMed
[15]
Yong-Jun Cho,
Sung-Min Cho, Seung-Hoon Sheen, et al. “Simple decompression of the ulnar nerve
for cubital tunnel syndrome.” J Korean Neurosurg Soc, vol. 42, no. 5,
pp. 382-387, 2007. View at: Publisher Site | PubMed
[16]
Malik
Jakirlic, Sanela Salihagic, Nedim Katica, et al. “Compression of Ulnar Nerve by
Ganglion Cyst in Guyon's Canal-a Case Report.” Acta Inform Med, vol. 31,
no. 4, pp. 326-328, 2023. View at: Publisher Site | PubMed
[17]
Kenan Güvenç,
Erdem Ateş, Zeynel Mert Asfuroğlu “Factors affecting functional outcomes after surgery to repair extensive
volar forearm lacerations with nerve injuries identified via quantitative and
qualitative methods.” Jt Dis Relat Surg, vol. 34, no. 2, pp. 405-412, 2023. View at: Publisher Site | PubMed
[18]
Dorcas E
Beaton, James G Wright, Jeffrey N Katz “Development of the QuickDASH:
comparison of three item-reduction approaches.” J Bone Joint Surg Am,
vol. 87, no. 5, pp. 1038-1046, 2005. View at: Publisher Site | PubMed
[19]
Abdus S
Burahee, Andrew D Sanders, Colin Shirley, et al. “Cubital tunnel syndrome.” EFORT
Open Rev, vol. 6, no. 9, pp. 743-750, 2021. View at: Publisher Site | PubMed
[20]
Arjun Ajith
Naik, Akshdeep Bawa, Anand Arya, et al. “Nerve entrapment around elbow.” J
Clin Orthop Trauma, vol. 19, pp. 209-215, 2021. View at: Publisher Site | PubMed
[21]
Veronica
Macchi, Cesare Tiengo, Andrea Porzionato, et al. “The cubital tunnel: a
radiologic and histotopographic study.” J Anat, vol. 225, no. 2, pp.
262-269, 2014. View at: Publisher Site | PubMed
[22]
Tianliang
Wang, Yang Wang, Chaoqun Yuan, et al. “Comparative Analysis of
Ultrasound-Assisted Precise Localization vs. Traditional Open Incision in Situ
Decompression for the Treatment of Cubital Tunnel Syndrome.” Turk Neurosurg,
vol. 34, no. 2, pp. 308-313, 2024. View at: Publisher Site | PubMed
[23]
Michael J Ho,
Ulrike Held, Klaus Steigmiller, et al. “Comparison of electrodiagnosis,
neurosonography and MR neurography in localization of ulnar neuropathy at the
elbow.” J Neuroradiol, vol. 49, no. 1, pp. 9-16, 2022. View at: Publisher Site | PubMed
[24]
Sophia Xirou,
Evangelos Anagnostou “Electrodiagnosis and Ultrasound Imaging for Ulnar Nerve
Entrapment at the Elbow: A Review.” Neurodiagn J, vol. 64, no. 4, pp.
175-192, 2024. View at: Publisher Site | PubMed
[25]
Kesha
Meghashyam, Mahesh Prakash, Tarun Narang, et al. “Role of shear wave
elastography in treatment follow-up of leprosy neuropathy.” J Ultrasound,
vol. 25, no. 2, pp. 265-272, 2022. View at: Publisher Site | PubMed
[26]
Yao Zhang,
Wei-Yao Liu, Wei-Li Xue, et al. “Diagnostic efficiency of conventional
ultrasound, shear wave elastography, and superb microvascular imaging in
evaluating ulnar neuropathy at the elbow.” Muscle Nerve, vol. 70, no. 2,
pp. 210-216, 2024. View at: Publisher Site | PubMed
[27]
Dominika Ślósarz, Elżbieta Poniewierka, Katarzyna Neubauer, et al. “Ultrasound Elastography in the Assessment of the
Intestinal Changes in Inflammatory Bowel Disease-Systematic Review.” J Clin Med, vol. 10, no. 18, pp. 4044,
2021. View at: Publisher Site | PubMed
[28]
Zhengyang Han,
Yuanjing Huang, Honghu Wang, et al. “Multimodal ultrasound imaging: A method to
improve the accuracy of diagnosing thyroid TI-RADS 4 nodules.” J Clin
Ultrasound, vol. 50, no. 9, pp. 1345-1352, 2022. View at: Publisher Site | PubMed
[29]
Jiaan Zhu, Li
Qiu, Dean Ta, et al. “Chinese Ultrasound Doctors Association Guideline on
Operational Standards for 2-D Shear Wave Elastography Examination of
Musculoskeletal Tissues.” Ultrasound Med Biol, vol. 50, no. 2, pp.
175-183, 2024. View at: Publisher Site | PubMed
[30]
Olga
Sánchez-Ramírez, Jaime Almazán-Polo, Ángel González-de-la-Flor “Reliability of
Ultrasound and Shear Wave Elastography in Assessing Lower Extremity Nerve
Stiffness and Excursion: A Systematic Review and Meta-Analysis.” Ultrasound
Med Biol, vol. 17, pp. S0301-5629(25)00085-7, 2025. View at: Publisher Site | PubMed
[31]
Thomas John
Carroll, Alexander Chirokikh, Julie Thon, et al. “Diagnosis of Ulnar Neuropathy
at the Elbow Using Ultrasound - A Comparison to Electrophysiologic Studies.” J
Hand Surg Am, vol. 48, no. 12, pp. 1229-1235, 2023. View at: Publisher Site | PubMed
[32]
Alexander
Alexei Chirokikh, Thomas John Carroll, Samantha Hoffman, et al. Where Does
Ultrasound Fit in the Diagnostic Algorithm for Cubital Tunnel Syndrome? Hand
(N Y), vol. 20, no. 1, pp. 87-91, 2023. View at: Publisher Site | PubMed
[33]
Ranjit Kumar
Chaudhary, Nikitha Karkala, Pankaj Nepal, et al. “Multimodality imaging review
of ulnar nerve pathologies.” Neuroradiol J, vol. 37, no. 2, pp. 137-151,
2024. View at: Publisher Site | PubMed
[34]
Kamal Mezian,
Jakub Jačisko, Radek Kaiser, et al. “Ulnar Neuropathy at the Elbow: From
Ultrasound Scanning to Treatment.” Front Neurol, vol. 12, pp. 661441,
2021. View at: Publisher Site | PubMed
[35]
Tiago Neto,
Johanna Johannsson, Ricardo J “Andrade Using ultrasound shear wave elastography
to characterize peripheral nerve mechanics: a systematic review on the
normative reference values in healthy individuals.” Ultrasonography,
vol. 43, no. 3, pp. 169-178, 2024. View at: Publisher Site | PubMed
[36]
David M
Rempel, Edward Diao “Entrapment neuropathies: pathophysiology and
pathogenesis.” [J Electromyogr Kinesiol, vol. 14, no. 1, pp. 71-75,
2004. View at: Publisher Site | PubMed
[37]
Ł Paluch, B H
Noszczyk, J Walecki, et al. “Shear-wave elastography in the diagnosis of ulnar
tunnel syndrome.” J Plast Reconstr Aesthet Surg, vol. 71, no. 11, pp.
1593-1599, 2018. View at: Publisher Site | PubMed
[38]
Andrea
Celli, Angelo De Crescenzo, Biagio Abate, et al. “Causes, symptoms, and
treatments of nerve entrapments around the elbow: Current concepts.” J
ISAKOS, vol. 9, no. 2, pp. 240-249, 2024. View at: Publisher Site | PubMed
