Received: Fri 21, Feb 2025
Accepted: Tue 18, Mar 2025
Abstract
Background: We aimed to identify the potential risk factors for early postoperative hemorrhage in patients undergoing endoscopic retrograde cholangiopancreatography (ERCP) with biliary stent placement for acute cholangitis.
Methods: A retrospective cohort study was conducted including 150 patients undergoing ERCP with biliary stent placement at Yangpu Hospital from January 2017 to December 2020 as training set, and followed up for six months to assess postoperative complications. The least absolute shrinkage and selection operator and multivariate logistic regression were conducted to identify independent risk factors associated with early postoperative hemorrhage, and a dynamic nomogram model was constructed. Another 40 patients treated between January and December 2023 were used for external validation. Model prediction performance was evaluated using the receiver operating characteristic (ROC) curve, calibration plot, and decision curve analysis.
Results: Inflammatory and liver function indicators recovered rapidly after operation. The incidence of early postoperative hemorrhage, defined as hemoglobin (Hb) drop exceeding 20.0 g/L, was 26.0% in the training set and 12.5% in the validation set. Multivariate analysis identified the proportion of neutrophils, presence of pancreatitis, TBIL and baseline Hb levels as significant predictors of early hemorrhage. The area under the curve under the ROC curves was 0.848 in the training set, and 0.846 in the validation set, which demonstrated a good prediction capability.
Conclusion: Preoperative presence of pancreatitis, TBIL, and inflammation serve as independent predictors of postoperative hemorrhage risk in patients undergoing ERCP with biliary stent placement.
Keywords
Biliary stent, choledocholithiasis, ERCP, hemorrhage, pancreatitis, risk factors
1. Introduction
Choledocholithiasis is the most prevalent etiology of biliary obstruction [1], leading to severe complications such as acute cholangitis, a life-threatening condition characterized by bacterial infection and inflammation of biliary tree, necessitating prompt intervention [2]. Endoscopic therapies, particularly endoscopic retrograde cholangiopancreatography (ERCP) with biliary stent placement, are established as the first-line treatment for acute cholangitis. ERCP with biliary stent placement is not only the gold standard for managing malignant biliary obstruction but also widely utilized for choledocholithiasis [3].
Meanwhile, ERCP with biliary stent placement is associated with significant complications, including post-procedural pancreatitis [4], perforation [5], and hemorrhage [6]. Among these, postoperative hemorrhage is one of the most severe adverse events, with incidence rates as high as 27% [7]. Recent studies indicate a decline in postoperative mortality but a concurrent increase in hemorrhage rates [8].
Previous research considered exogenous risk factors, such as precut sphincterotomy and endoscopic sphincterotomy, are the risk factors for postoperative hemorrhage [7, 9]. Also, intraprocedural visible bleeding was a potential predictor [10]. However, endogenous factors, which may play a key role in postoperative hemorrhage, remain poorly understood [11].
Post-ERCP hemorrhage is defined as hematemesis and/or melena, or a hemoglobin (Hb) drop exceeding 20.0 g/L. The postoperative decline in Hb levels is an objective marker of intraoperative blood loss and correlates more closely with actual blood loss than subjective estimates. Early Hb reduction is indeed a marker of the likelihood of significant injury, and indicates a poor prognosis after surgery [12]. Furthermore, the degree of Hb decline has been identified an independent risk factor for postoperative complications [13].
Hence, novel biomarkers predicting hemorrhage are urgently needed to improve post-ERCP clinical outcomes and reduce mortality rates. Therefore, this study aimed to explore the early hemorrhage after endoscopic biliary stent placement in patients with acute cholangitis, elucidate possible factors related to the early hemorrhage, and, eventually, design a prediction model that would provide a valuable evaluation tool for preventing bleeding in patients with endoscopic biliary stent placement.
2. Materials and Methods
2.1. Study Design
This was a retrospective study on patients undergoing ERCP with biliary stent placement for acute cholangitis conducted by the Department of General Surgery (Yangpu Hospital, Tongji University School). A total of 150 patients from January 2017 and December 2020 were selected as the training set, and 40 patients from January to December 2023 were selected as the validation set. The study involving human participants was reviewed and approved by the ethics committee of Yangpu Hospital (LL-2021-SCI-007). All selected patients were inpatients, and the discharged patients were followed up for six months telephonically and in the clinic. The inclusion criteria were as follows: i) patients aged > 18 years; and ii) patients with choledocholithiasis who voluntarily provided signed informed consent for the ERCP treatment. The exclusion criteria were as follows: i) patients with serious underlying diseases such as malignant tumors, renal failure, and liver cirrhosis, which could affect biochemical indicators in the blood; ii) patients with incomplete medical records; and iii) patients lost to follow-up.
2.2. ERCP Procedure
The procedures were performed by two experienced endoscopic physicians (Wang and Pei) who conducted annual operations on more than 200 patients. Analgesic (pethidine) and sedative (diazepam) drugs were given to patients before the operation. Biliary stenting was performed using standard duodenoscopes (Olympus Medical Systems, PA, USA). Two types (8.5F, 9cm; 7F, 14cm) of biliary stents were mainly used. The standard of successful operation was successful biliary stent placement and reconstruction of the bile drainage channel. After ERCP, the patients were fasted and given intravenous nutrient rehydration. The blood samples were routinely taken 6 and 24 h after ERCP for a routine blood test and to detect biochemical index and amylase and lipase activities. All patients were monitored for at least 48 h to prevent possible ERCP-related acute complications.
2.3. Observation Indicators
The observation indicators included age, sex, hospitalization, preoperative laboratory examinations such as white blood cell (WBC), hemoglobin (Hb), platelet, C-reactive protein (CRP), proportion of neutrophils, total bilirubin (TBIL), alanine transaminase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), creatinine, prothrombin time, fibrinogen, D-dimer, and comorbidities such as hypertension, diabetes, pancreatitis. Postoperative laboratory examinations were conducted 24 h after ERCP.
2.4. Statistical Analysis
The data were obtained by checking the medical system of the hospital and reviewing the corresponding case records, laboratory tests, endoscopic reports, and other corresponding examination results. The analyses were performed using SPSS version 25.0 (IBM Corp., NY, USA) and R software (version 4.0.2). The compatibility of variables was investigated using the Shapiro-Wilks test. The characteristics of patients were expressed using descriptive statistics. Continuous variables were presented as mean with standard deviation or median with interquartile spacing and compared with student t-test or Mann-Whitney U test. Categorical variables were compared using the chi-square test. All statistical test were 2-sided, and P value < 0.05 indicated a statistically significant difference.
Least absolute shrinkage and selection operation (LASSO) regression analysis was used to choose non-zero coefficients that best predict early hemorrhage risk in patients undergo ERCP with endoscopic biliary stent placement in the training set. Then, multivariable logistic regression was performed to determine the final predictors. Based on multivariate logistic regression analysis, a dynamic nomogram was constructed to predict the probability of early hemorrhage. Another 40 patients were selected for external validation. A receiver operating characteristic (ROC) curve and a calibration curve were used to assess the early hemorrhage risk nomogram. Decision curve analysis (DCA) was performed to assess the clinical applicability of the early hemorrhage risk model.
3. Results
A total of 190 patients who underwent ERCP with biliary stent placement were treated and monitored at Yangpu Hospital, School of Medicine, Tongji University, including 150 in the training set and 40 in the validation set. During the six-month follow-up, no serious surgical complications were observed. The incidence of postoperative hemorrhage was 26.0% in the training set and 12.5% in the validation set. The blood samples were examined to investigate the efficacy of ERCP with biliary stent placement in the training set. ERCP with biliary stent placement exhibited significant reductions in WBC count (P = 0.006), the proportion of neutrophils (P = 0.009), TBIL level (P < 0.001), as well as the levels of ALT and AST (both P < 0.001, Table 1).
TABLE 1: Comparison of laboratory findings after ERCP
with biliary stent placement in the training set.
|
Variable |
preoperative laboratory findings |
postoperative laboratory findings |
Z value |
P value |
|
WBC (*109/L) |
9.6 (6.9-13.1) |
8.3 (6.2-11.9) |
-2.758 |
0.006 |
|
Proportion of neutrophils (%) |
85.8 (74.2-91.4) |
83.0(75.0-88.3) |
-2.611 |
0.009 |
|
TBIL (μmol/L) |
55.9 (25.3-98.5) |
32.7 (19.4-62.9) |
-6.449 |
< 0.001 |
|
ALT (μ/L) |
137.0 (70.3-248.5) |
73.5 (42.8-139.8) |
-8.325 |
< 0.001 |
|
AST (μ/L) |
139.5 (59.8-294.0) |
47.0 (28.8-86.0) |
-8.974 |
< 0.001 |
WBC: White Blood Cell; CRP: C-Reactive Protein; TBIL: Total Bilirubin; ALT: Alanine Transaminase; AST: Aspartate Aminotransferase.
Based on the data from 150 patients in the training set, after conducting LASSO regression with five-fold cross-validation, 20 sociodemographic and clinical characteristics were reduced to 10 potential predictors with non-zero coefficients. The pathway of variable shrinkage and cross-validation was illustrated in (Figure 1). The LASSO analysis showed that early hemorrhage was significantly associated with baseline Hb levels, the proportion of neutrophils, TBIL levels, and the presence of pancreatitis.
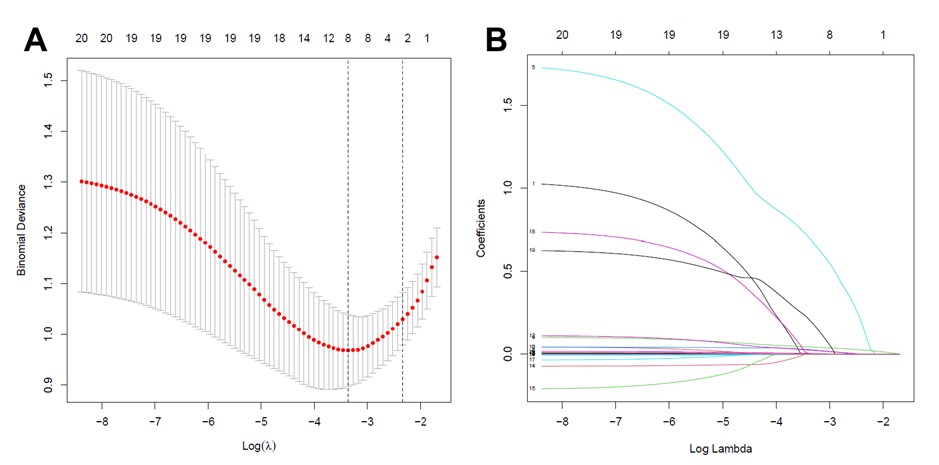
LASSO: Least Absolute Shrinkage and Selection Operator; SE: Standard Error.
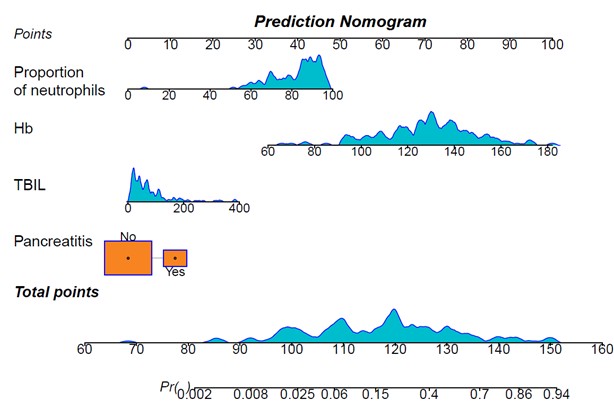
Based on these findings, we conducted a multiple logistic regression analysis. The results showed that four baseline variables, including the proportion of neutrophils [odds ratio (OR) = 1.072, 95% confidence interval (CI) = 1.017-1.137, P = 0.014], presence of pancreatitis (OR = 3.314, 95% CI = 1.048-10.929, P = 0.043), TBIL levels (OR = 1.012, 95% CI = 1.004-1.019, P = 0.002), and baseline Hb levels (OR = 1.069, 95% CI = 1.038-1.107, P < 0.001) were identified to predict early hemorrhage risk in ERCP with biliary stent placement patients (Table 2). The nomogram in (Figure 2) visually represents the results of the logistic regression analysis.
TABLE 2: Multivariate logistic regression
analysis of predictors selected through LASSO regression in the training set.
|
Variable |
OR (95%CI) |
P value |
|
WBC (*109/L) |
1.056 (0.954-1.169) |
0.292 |
|
Hb (g/L) |
1.069 (1.038-1.107) |
<0.001 |
|
CRP (mg/L) |
0.993 (0.983-1.002) |
0.128 |
|
proportion of neutrophils (%) |
1.072 (1.017-1.137) |
0.014 |
|
TBIL (μmol/L) |
1.012 (1.004-1.019) |
0.002 |
|
ALT (μ/L) |
1.001 (0.999-1.003) |
0.370 |
|
PT (s) |
0.82 (0.564-1.135) |
0.263 |
|
pancreatitis |
3.314 (1.048-10.929) |
0.043 |
|
diabetes |
1.771 (0.583-5.402) |
0.309 |
|
hypertension |
1.835 (0.672-5.201) |
0.240 |
WBC: White Blood Cell; Hb: Hemoglobin; CRP: C-reactive protein; TBIL: total bilirubin; ALT: Alanine Transaminase; PT: Prothrombin Time.
There were no significant differences observed in terms of the predicted variables between the training set and the validation set (Table 3). Model discrimination ability was assessed by calculating the area under the curve (AUC) of the ROC curve. As shown in (Figure 3A), the AUC of the training set was 0.848 (95% CI = 0.780-0.916), while in (Figure 3B), the AUC of the validation cohort was 0.846 (95% CI = 0.703-0.989), which demonstrated a good predictive performance. The calibration curves of the nomogram illustrated excellent agreement between the predicted probabilities of early hemorrhage and the actual observations in the training set (Figure 3C). Furthermore, the nomogram net benefit rate (the red line) was found to be above both the All and the None lines using DCA, indicating that the model had strong clinical practicability (Figure 3D).
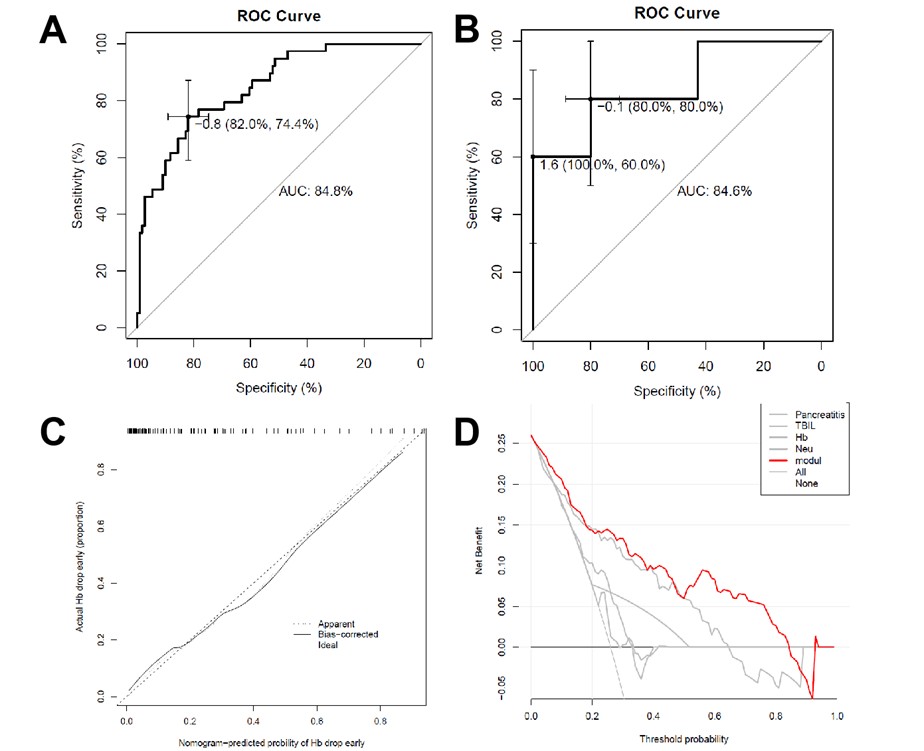
TABLE 3: Demographics and clinical
characteristics of training and validation sets.
|
Variable |
Test Group (N = 150) |
Validation Group (N = 40) |
P value |
|
Hb (g/L) |
128 (20.9) |
132 (17.8) |
0.187 |
|
proportion of neutrophils (%) |
82.0 (12.6) |
80.2 (12.2) |
0.408 |
|
TBIL (μmol/L) |
77.2 (74.3) |
69.3 (46.6) |
0.412 |
|
pancreatitis (%) |
29 (19.3%) |
12 (30.0%) |
0.215 |
Hb: Hemoglobin; TBIL: Total Bilirubin.
4. Discussion
This study examined 190 acute cholangitis patients who underwent ERCP with biliary stent placement at Yangpu Hospital. The cohort was divided into a training set (n=150) demonstrating a postoperative hemorrhage prevalence of 26.0% and a validation set (n=40) with a hemorrhage prevalence of 12.5%. A machine learning derived model was developed and validated to assess early hemorrhage risk in these patients. The model construction incorporated LASSO regression for feature selection followed by multiple logistic regression analysis, ultimately identifying four significant baseline predictors: the proportion of neutrophils, presence of pancreatitis, TBIL and baseline Hb levels. The resulting nomogram from this model provides clinicians with a visual representation to estimate early hemorrhage in acute cholangitis patients undergoing ERCP with biliary stent placement.
ERCP with biliary stent placement has been established as a safe and effective treatment for acute cholangitis [14], especially in patients involving biliary obstruction secondary to common bile duct stones [15]. Invasive operation is undoubtedly a great risk for frail elderly patients having multiple comorbidities. Especially in the state of systemic inflammatory stress, endoscopic surgery is relatively minimally invasive, safe, and effective [16]. Meanwhile, recent evidence indicates that biliary sphincterotomy is not required for bile duct stent placement [17], which further minimizing procedural trauma and enhancing patient safety. In the present study, follow-up over six months revealed no incidence of serious adverse events, with patients demonstrating significant improvement in inflammatory and liver function parameters after surgery (Table 1).
Postoperative hemorrhage remains one of the most critical complications following ERCP. In the present study, despite the exclusion of traumatic factors, the incident of postoperative hemorrhage, defined as Hb drop exceeding 20.0 g/L, was still high at 26.0% in the training set and 12.5% in the validation set. Previous research showed that an early drop in Hb serves as a marker for significant injury risk [18, 19]. The clinical significance of Hb decline extends beyond its immediate effects, as it compromises tissue oxygen delivery, potentially leading to poor postoperative prognosis [12]. Reducing operative blood loss improves patient outcomes and reduces healthcare costs [20]. Supporting evidence from gastric cancer surgery indicates that the magnitude of postoperative Hb reduction constitutes an independent risk factor for surgical complications, with the nadir postoperative Hb level and requirement for blood transfusion emerging as particularly significant predictors of adverse outcomes [13]. Similar results were found in patients after percutaneous coronary intervention [21] and those with acute coronary syndromes [22].
Biliary stone accounts for 30%–70% of acute pancreatitis (AP) [23]. The pathogenesis of biliary pancreatitis may be due to the migration of gallstones to affect the main pancreatic duct, resulting in intracranial high pressure accompanied by defective pancreatic secretion elimination and regurgitation [24]. Hemorrhage is an infrequent but potentially fatal complication of AP, and it can increase the risk of mortality and the length of hospital stay in patients with AP [25]. Gastrointestinal bleeding is considered an essential form of pancreatitis related bleeding [26]. Previous studies have shown that 50%–65% of patients with AP may have acute gastrointestinal mucosal lesions [27]; especially in the state of AP and acute cholangitis co-occurrence, stress ulcer–related bleeding is particularly prone to occur. Previous studies have shown bleeding to be more common in infected necrosis compared with sterile necrosis [28]. Besides, edema of the digestive tract with submucosal bleeding due to inflammation and entering the duodenum with a duodenoscope may cause bleeding in the digestive tract. Besides gastrointestinal hemorrhage, vascular lesions and vessel rupture caused by necrosis factors associated with pancreatitis are also considered to be the causes of pancreatitis-associated hemorrhage [28]. In our study, pancreatitis (OR = 3.314, P = 0.043) was supposed to be an essential factor in postoperative hemorrhage.
Acute Hb reduction is a common phenomenon in acute infection, mainly due to a decrease in the production of erythropoietin due to the release of inflammatory cytokines, such as tumor necrosis factor, interleukin-1 (IL-1), and IL-6, as well as increased destruction of red cells due to hemolysis and bleeding [29]. In the present study, the proportion of neutrophils (OR = 1.072, P = 0.014), which reflects the level of inflammation, were identified to predict an early drop in Hb. As the principal component of erythrocytes, Hb levels provide valuable insights into the degree of erythrocyte damage. Multiple organ dysfunction resulting from acute cholangitis was associated with the presence of high levels of oxidative stress [30], which could induce erythrocyte and Hb damage via altering the structure and function of Hb, causing the denaturation and precipitation of Hb, or directly penetrating the red blood cell membrane and oxidizing Hb to ferric Hb. An increase in lipid peroxidation has been noted as evidence of oxidative damage in red blood cells [31]. After ERCP intervention, the antioxidant capacity of red blood cells significantly improved and returned to normal [32]. This finding is further supported by experimental evidence showing reduced blood loss in rats after the surgery following antioxidant administration [33]. Collectively, these findings strongly implicate inflammation status as a critical determinant of early hemorrhage after the surgery.
The obstruction of the biliary tree and the inability to excrete bile into the intestines lead to the continuous accumulation of bile. Hence, large amounts of endotoxins are produced, which can not only damage platelets and vascular endothelium [34], while also cause systemic inflammatory response syndrome, multiple organ dysfunction syndrome [35], and sepsis [36]. Patients with obstructive jaundice were found to have an increased bleeding tendency [34], this occurs paradoxically in the presence of normal platelet counts. Notably, the thromboelastographic parameter, maximum amplitude value, which represents the strength of clots, was observed to be higher than normal. Meanwhile, the mean prothrombin time was significantly higher in patients with jaundice [37]. The tissue factor pathway has been identified as a critical role in the appeal phenomenon [36]. Increased levels of oxidative stress were found in patients with obstructive jaundice [30] and animals [38, 39]. Antioxidative and redox systems play more important roles in maintaining the homeostasis of erythrocytes against oxidative insult compared with ordinary cells. Hence, erythrocytes are a preferential target of oxidative stress in the blood [40]. The present study also find an association between TBIL (P = 0.002) and postoperative bleeding, further emphasizing the complex interplay between biliary obstruction and coagulation function.
This study has several limitations inherent to its design as a single-center retrospective analysis. The data only from a single institution may limit the generalizability of our findings. The relatively modest sample size may reduce the statistical power of our analyses. Furthermore, certain confounding variables could not be controlled, and retrospectively supplementing incomplete data was difficult, thus affecting the research results.
5. Conclusion
Endoscopic stent placement for biliary drainage is a safe and effective treatment for acute cholangitis due to biliary obstruction. A significant drop in Hb after ERCP was observed in this study. Preoperative presence of pancreatitis, TBIL, and inflammation serve as independent predictors of postoperative hemorrhage risk in patients undergoing ERCP with biliary stent placement.
Data Availability
All data generated or analyzed during this study are included in this published article.
Conflicts of Interest
None.
Funding
This study was supported by Shanghai Municipal Health Commission Clinical Research Program (No. 20194Y0457 to Erli Pei), and Shanghai Municipal Health Commission Policy Program (No. 2022HP70 to Li Cai).
Acknowledgments
Not applicable.
Ethics Approval and Consent to Participate
The study involving human participants was reviewed and approved by the ethics committee of Yangpu Hospital (LL-2021-SCI-007). Our research was conducted without any human experimentation or the collection of tissue samples. All data were rigorously anonymized.
Clinical Trial Number
Not applicable.
REFERENCES
[1]
Amir Houshang
Mohammad Alizadeh “Cholangitis: Diagnosis, Treatment and Prognosis.” J Clin
Transl Hepatol, vol. 5, no. 4, pp. 404-413, 2017. View at: Publisher Site | PubMed
[2]
David Lan
Cheong Wah, Christopher Christophi, Vijayaragavan Muralidharan “Acute
cholangitis: current concepts.” ANZ J Surg, vol. 87, no. 7-8, pp.
554-559, 2017. View at: Publisher Site | PubMed
[3]
J-M Dumonceau,
A Tringali, D Blero, et al. “European Society of Gastrointestinal Endoscopy.
Biliary stenting: indications, choice of stents and results: European Society
of Gastrointestinal Endoscopy (ESGE) clinical guideline.” Endoscopy,
vol. 44, no. 3, pp. 277-298, 2012. View at: Publisher Site | PubMed
[4]
Yegor
Tryliskyy, Gavin J Bryce “Post-ERCP pancreatitis: Pathophysiology, early
identification and risk stratification.” Adv Clin Exp Med, vol. 27, no.
1, pp. 149-154, 2018. View at: Publisher Site | PubMed
[5]
Guiying Zhu,
Fenglin Hu, Changmiao Wang “Recent advances in prevention and management of
endoscopic retrograde cholangiopancreatography-related duodenal perforation.” Wideochir
Inne Tech Maloinwazyjne, vol. 16, no. 1, pp. 19-29, 2021. View at: Publisher Site | PubMed
[6]
Rupjyoti
Talukdar “Complications of ERCP.” Best Pract Res Clin Gastroenterol,
vol. 30, no. 5, pp. 793-805, 2016. View at: Publisher Site | PubMed
[7]
Jing Yan,
Chun-Xia Zhou, Chong Wang, et al. “Risk factors for delayed hemorrhage after
endoscopic sphincterotomy.” Hepatobiliary Pancreat Dis Int, vol. 19, no.
5, pp. 467-472, 2020. View at: Publisher Site | PubMed
[8]
Faiz Afridi,
Laura Rotundo, Mirela Feurdean, et al. “Trends in Post-Therapeutic Endoscopic
Retrograde Cholangiopancreatography Gastrointestinal Hemorrhage, Perforation
and Mortality from 2000 to 2012: A Nationwide Study.” Digestion, vol.
100, no. 2, pp. 100-108, 2019. View at: Publisher Site | PubMed
[9]
Xiaoling Ye,
Yu Zhang, Xinyue Wan, et al. “Analysis of Risk Factors in Endoscopic Retrograde
Cholangiopancreatography-Related Immediate and Delayed Hemorrhage.” Dig Dis
Sci, vol. 66, no. 12, pp. 4467-4474, 2021. View at: Publisher Site | PubMed
[10]
E J Williams,
S Taylor, P Fairclough, et al. “Risk factors for complication following ERCP;
results of a large-scale, prospective multicenter study.” Endoscopy,
vol. 39, no. 9, pp. 793-801, 2007. View at: Publisher Site | PubMed
[11]
Nicholas M
Szary, Firas H Al-Kawas “Complications of endoscopic retrograde
cholangiopancreatography: how to avoid and manage them.” Gastroenterol
Hepatol (N Y), vol. 9, no. 8, pp. 496-504, 2013. View at: PubMed
[12]
Li Zhou,
Xinhao Liu, Min Yan, et al. “Postoperative Nadir Hemoglobin and Adverse
Outcomes in Patients Undergoing On-Pump Cardiac Operation.” Ann Thorac Surg,
vol. 112, no. 3, pp. 708-716, 2021. View at: Publisher Site | PubMed
[13]
Do-Hyun Jung,
Hyuk-Joon Lee, Dong-Seok Han, et al. “Impact of perioperative hemoglobin levels
on postoperative outcomes in gastric cancer surgery.” Gastric Cancer,
vol. 16, no. 3, pp. 377-382, 2013. View at: Publisher Site | PubMed
[14]
Ru-ling Zhang,
Li Cheng, Xiao-bo Cai, et al. “Comparison of the safety and effectiveness of
endoscopic biliary decompression by nasobiliary catheter and plastic stent
placement in acute obstructive cholangitis.” Swiss Med Wkly, vol. 143,
pp. w13823, 2013. View at: Publisher Site | PubMed
[15]
Eoin Slattery,
Vikrant Kale, Waqas Anwar, et al. “Role of long-term biliary stenting in
choledocholithiasis.” Dig Endosc, vol. 25, no. 4, pp. 440-443, 2013.
View at: Publisher Site | PubMed
[16]
Gen Tohda,
Masahiro Ohtani, Masaki Dochin “Efficacy and safety of emergency endoscopic
retrograde cholangiopancreatography for acute cholangitis in the elderly.” World
J Gastroenterol, vol. 22, no. 37, pp. 8382-8388, 2016. View at: Publisher Site | PubMed
[17]
C Mel Wilcox,
Hwasoon Kim, Jayapal Ramesh, et al. “Biliary sphincterotomy is not required for
bile duct stent placement.” Dig Endosc, vol. 26, no. 1, pp. 87-92, 2014.
View at: Publisher Site | PubMed
[18]
Alparslan
Turan, Barak Cohen, Eva Rivas, et al. “Association between postoperative
haemoglobin and myocardial injury after noncardiac surgery: a retrospective
cohort analysis.” Br J Anaesth, vol. 126, no. 1, pp. 94-101, 2021. View
at: Publisher Site | PubMed
[19]
Yash P
Chaudhry, Aoife MacMahon, Sandesh S Rao, et al. “Predictors and Outcomes of
Postoperative Hemoglobin of <8 g/dL in Total Joint Arthroplasty.” J Bone
Joint Surg Am, vol. 104, no. 2, pp. 166-171, 2022. View at: Publisher Site | PubMed
[20]
A Shah, A J R
Palmer, A A Klein “Strategies to minimize intraoperative blood loss during
major surgery.” Br J Surg, vol. 107, no. 2, pp. e26-e38, 2020. View at: Publisher Site | PubMed
[21]
Toshiki Kuno,
Yohei Numasawa, Takahisa Mikami, et al. “Association of decreasing hemoglobin
levels with the incidence of acute kidney injury after percutaneous coronary
intervention: a prospective multi-center study.” Heart Vessels, vol. 36,
no. 3, pp. 330-336, 2021. View at: Publisher Site | PubMed
[22]
Gjin Ndrepepa,
Franz-Josef Neumann, Maurizio Menichelli, et al. “Prognostic value of
haemoglobin drop in patients with acute coronary syndromes.” Eur J Clin
Invest, vol. 51, no. 12, pp. e13670, 2021. View at: Publisher Site | PubMed
[23]
Hsiu-Nien
Shen, Wen-Ching Wang, Chin-Li Lu, et al. “Effects of gender on severity,
management and outcome in acute biliary pancreatitis.” PLoS One, vol. 8,
no. 2, pp. e57504, 2013. View at: Publisher Site | PubMed
[24]
Philippe Lévy,
Arnaud Boruchowicz, Patrick Hastier, et al. “Diagnostic criteria in predicting
a biliary origin of acute pancreatitis in the era of endoscopic ultrasound:
multicentre prospective evaluation of 213 patients.” Pancreatology, vol.
5, no. 4-5, pp. 450-456, 2005. View at: Publisher Site | PubMed
[25]
Hsiu-Nien
Shen, Chin-Li Lu, Chung-Yi Li “The effect of gastrointestinal bleeding on
outcomes of patients with acute pancreatitis: a national population-based
study.” Pancreatology, vol. 12, no. 4, pp. 331-336, 2012. View at: Publisher Site | PubMed
[26]
Surinder S
Rana, Vishal Sharma, Deepak K Bhasin, et al. “Gastrointestinal bleeding in
acute pancreatitis: etiology, clinical features, risk factors and outcome.” Trop
Gastroenterol, vol. 36, no. 1, pp. 31-35, 2015. View at: Publisher Site | PubMed
[27]
Tai-An Chen,
Gin-Ho Lo, Chiun-Ku Lin, et al. “Acute pancreatitis-associated acute
gastrointestinal mucosal lesions: incidence, characteristics, and clinical
significance.” J Clin Gastroenterol, vol. 41, no. 6, pp. 630-634, 2007.
View at: Publisher Site | PubMed
[28]
Praveen Kumar
Sharma, Kaushal Madan, Pramod Kumar Garg “Hemorrhage in acute pancreatitis:
should gastrointestinal bleeding be considered an organ failure?” Pancreas,
vol. 36, no. 2, pp. 141-145, 2008. View at: Publisher Site | PubMed
[29]
Gassan Fuad
Muady, Haim Bitterman, Arie Laor, et al. “Hemoglobin levels and blood
transfusion in patients with sepsis in Internal Medicine Departments.” BMC
Infect Dis, vol. 16, no. 1, pp. 569, 2016. View at: Publisher Site | PubMed
[30]
Stelios F
Assimakopoulos, Konstantinos Grintzalis, Konstantinos C Thomopoulos, et al.
“Plasma superoxide radical in jaundiced patients and role of xanthine oxidase.”
Am J Med Sci, vol. 336, no. 3, pp. 230-236, 2008. View at: Publisher Site | PubMed
[31]
Tao Yuan,
Wen-Bin Fan, Yu Cong, et al. “Linoleic acid induces red blood cells and
hemoglobin damage via oxidative mechanism.” Int J Clin Exp Pathol, vol.
8, no. 5, pp. 5044-5052, 2015. View at: PubMed
[32]
Ibrahim H
El-Sayed, Ibrahim M El kady, Gamal A Badra “The effect of endoscopic retrograde
cholangiopancreatography on the serum IL-18 and erythrocytes antioxidative
capacity in biliary obstructive jaundice.” Clin Chim Acta, vol. 336, no.
1-2, pp. 123-128, 2003. View at: Publisher Site | PubMed
[33]
Hong Qian, Tao
Yuan, Jian Tong, et al. “Antioxidants Attenuate Oxidative Stress-Induced Hidden
Blood Loss in Rats.” Turk J Haematol, vol. 34, no. 4, pp. 334-339, 2017.
View at: Publisher Site | PubMed
[34]
Tebessüm
Cakir, Asim Cingi, Cumhur Yeğen “Coagulation dynamics and platelet functions in
obstructive jaundiced patients.” J Gastroenterol Hepatol, vol. 24, no. 5,
pp. 748-751, 2009. View at: Publisher Site | PubMed
[35]
Efstathios T
Pavlidis, Theodoros E Pavlidis “Pathophysiological consequences of obstructive
jaundice and perioperative management.” Hepatobiliary Pancreat Dis Int,
vol. 17, no. 1, pp. 17-21, 2018. View at: Publisher Site | PubMed
[36]
Vassilios
Papadopoulos, Dimitrios Filippou, Evangelos Manolis, et al. “Haemostasis
impairment in patients with obstructive jaundice.” J Gastrointestin Liver
Dis, vol. 16, no. 2, pp. 177-186, 2007. View at: PubMed
[37]
Richard J
Egan, Jonathan Nicholls, Sarah Walker, et al. “Routine coagulation screening is
an unnecessary step prior to ERCP in patients without biochemical evidence of
jaundice: a cross-centre study.” Int J Surg, vol. 12, no. 11, pp.
1216-1220, 2014. View at: Publisher Site | PubMed
[38]
Shintaro
Fukushima, Hiroyasu Okuno, Nobuyuki Shibatani, et al. “Effect of biliary
obstruction and internal biliary drainage on hepatic cytochrome P450 isozymes
in rats.” World J Gastroenterol, vol. 14, no. 16, pp. 2556-2560, 2008.
View at: Publisher Site | PubMed
[39]
Ata Ahmet
Alturfan, Erman Aytaç, Ebru Emekli-Alturfan, et al. “Serum total sialic acid as
a novel complementary candidate marker of hepatic damage in obstructive
jaundice.” Ann Clin Lab Sci, vol. 44, no. 1, pp. 56-61, 2014. View at: PubMed
[40] Junichi Fujii, Takujiro Homma, Sho Kobayashi, et al. “Erythrocytes as a preferential target of oxidative stress in blood.” Free Radic Res, vol. 55, no. 5, pp. 562-580, 2021. View at: Publisher Site | PubMed
