Received: Mon 21, Apr 2025
Accepted: Fri 09, May 2025
Abstract
Background: The clinical outcomes of rheumatic mitral valve repair (MVP) have been controversial, particularly regarding the reoperation rates. Therefore, we conducted this meta-analysis to comprehensively and systematically evaluate clinical outcomes, focusing on reoperation rates.
Methods: PubMed, EMBASE, Web of Science, and Cochrane Library were searched for articles and abstracts published between January 1, 1990, and September 21, 2023, to compare the clinical outcomes of MVP and mitral valve replacement (MVR) in patients with rheumatic heart disease (RHD).
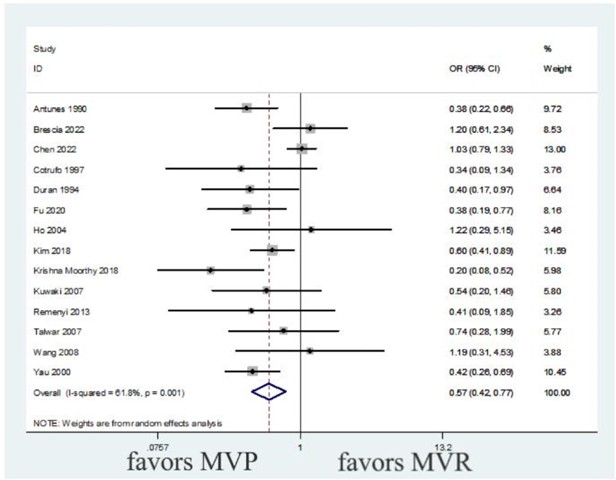
Results: After screening the titles and abstracts of 2703 articles, 165 articles were reviewed. Twenty articles met our inclusion criteria, the 20 included studies were observational studies, comprising 4492 MVP and 7913 MVR cases. MVP decreased early mortality (odds ratio (OR): 0.63, 95% CI: 0.50-0.78, P < 0.001) and long-term mortality (OR: 0.57, 95% CI: 0.42-0.77,P < 0.001) and reduced the rates of thromboembolism (OR: 0.58, 95% CI: 0.46-0.74, P < 0.001), bleeding (OR: 0.70, 95% CI: 0.55-0.89, P = 0.004), and heart failure (OR: 0.28, 95% CI: 0.12-0.67, P = 0.004). Infective endocarditis (P = 0.786), stroke (P = 0.503), and atrial fibrillation (P = 0.180) were not significantly different between the groups. To objectively analyze the reoperation rate, the studies were divided into three subgroups and analyzed according to the era of surgery. The risk of reoperation for MVP was high before 2000 (OR, 3.67; 95% CI: 1.98-6.78, P < 0.001). From 2000 to 2010, the risk of reoperation decreased but remained high overall. After 2010, the reoperation rate was similar to that of MVR.
Conclusion: In patients with RHD, MVP reduces early mortality, long-term mortality, thromboembolism, bleeding, and heart failure compared to MVR. Historically, MVP has had a higher reoperation rate than MVR, but in recent years, this rate has gradually declined. After 2010, there was no significant difference in reoperation rates between MVP and MVR.
Keywords
Rheumatic heart disease, mitral valve surgery, mitral valve repair, replacement, reoperation rate
1. Introduction
Rheumatic heart disease remains the most common manifestation of heart valve disease. It affects approximately 41 million people, resulting in more than 300,000 deaths and 10 million people annually. Middle- and low-income countries are particularly affected with high incidence and mortality rates [1, 2]. Rheumatic heart disease most commonly involves the mitral valve and manifests as mitral stenosis, regurgitation, or both [3]. The mainstays of treatment are percutaneous balloon mitral valvuloplasty and mitral valve surgery [4, 5]. However, percutaneous balloon mitral valvuloplasty has many limitations, such as moderate-to-severe mitral regurgitation, significant calcification of the mitral valve, and fresh thrombus in the left atrium [6]. Valve repair and replacement have become more common forms of treatment. It is well known that in severe degenerative mitral valve disease, repair has a significant advantage over replacement [7, 8], in rheumatic mitral valve disease, this view has been controversial [9, 10]. In recent years, the reoperation rates have been the focus of controversy. Therefore, a meta-analysis was performed to comprehensively and systematically evaluate the clinical outcomes, with a particular focus on reoperation rates.
2. 2. Methodology
The meta-analysis was registered with PROSPERO (under number CRD42024497774 ) and follows the guidelines outlined by the Preferred Reporting Items for Systematic Review and Meta-analysis (PRISMA, Supplemental Digital Content 1) [11], and the A Measurement Tool to Assess Systematic Reviews (AMSTAR 2) checklist (Supplemental Digital Content 2) [12].
2.1. Search Strategy
Four electronic databases (PubMed, EMBASE, Web of Science, and Cochrane Library) were searched to identify relevant articles and abstracts published between January 1, 1990, and September 21, 2023, to compare the clinical outcomes of MVP and MVR in patients with RHD. The keywords searched were (mitral valve) AND [(repair) OR (Annuloplasty) OR (reconstruction)] AND (replacement) AND [(Rheumatic) OR (Bouillaud's Disease) OR (RHD)].
2.2. Eligibility Criteria
Studies were included if they i) involved patients with RHD, ii) directly compared MVP and MVR, and iii) reported at least one clinical outcome. Studies were excluded if they i) involved patients without RHD; ii) focused exclusively on either MVP or MVR; or iii) were conference proceedings or reviews. Early mortality was defined as mortality from any cause during hospitalization or within 30 days after surgery. Long-term mortality was defined as death from any cause beyond 30 days after surgery or during follow-up. Reoperation was defined as another procedure that involved the mitral valve. Valve-related events included infective endocarditis, thromboembolism, and bleeding. Adverse events included heart failure, stroke, and atrial fibrillation. Heart failure was defined as a new postoperative heart failure or death due to heart failure. Stroke and atrial fibrillation were defined as new postoperative stroke and new atrial fibrillation after discharge, respectively.
2.3. Data Extraction
Two reviewers independently extracted and summarized the data according to a uniform format, first screened out duplicate or irrelevant studies by title and abstract, then reviewed the full text to confirm eligibility and extracted key data, including study author names, country of origin, patient statistics (intervention, year of surgery, sample size, age, sex, cardiac function, atrial fibrillation, type of valve replacement, characteristics of mitral lesions, major additional procedures), and results of the main outcome measures. For propensity score matching studies, data were extracted separately for before and after matching. Disagreements, if any, were resolved by discussion, with the involvement of a third reviewer when needed to reach consensus.
2.4. Risk of Bias and Quality of Evidence Assessment
The risk of bias of the included studies was assessed using the Risk of Bias in Non-Randomized Studies of Interventions tool (ROBINS-II) [13]. Two independent reviewers assessed the risk of bias for each study. In case of disagreement, a third reviewer reviewed the data and made the final risk of bias judgment.
2.5. Statistical Analysis
Stata 18.0 software was used to meta-analyze the data and draw the relevant graphs. Heterogeneity among independent studies was analyzed using the Q test and I² test, and if there was no heterogeneity among studies (I² ≤ 50% and P > 0.10 for the Q test), the combined OR value and its 95% confidence interval were calculated using a fixed-effects model. If heterogeneity was present, a random effects model was used. The stability of the combined values was explored by excluding the included studies individually to determine the sensitivity of the findings, and publication bias was analyzed using funnel plots, Begg's test, and Egger's test. Statistical significance was set at P < 0.05.
3. Results
3.1. Study Selection and Patients’ Characteristics
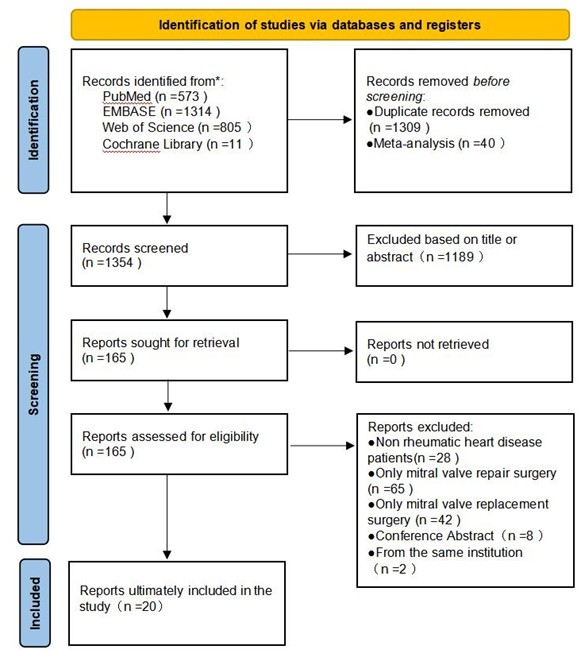
After screening the titles and abstracts of 2703 articles, a total of 165 articles were screened, of which 20 met our inclusion criteria [14-33], and the 20 included studies were observational studies. These articles comprised 4492 cases of MVP and 7913 cases of MVR. Following the PRISMA guidelines, the results of the literature search are shown in (Figure 1), a summary of the characteristics of individual studies is shown in (Table 1), and the prevalence of the main risk factors in individual studies is shown in (Table 2).
TABLE 1: Summary
of characteristics of individual studies.
|
Study |
Country |
Years of surgery |
Repair |
Replacement |
Mean age (years) |
Male/female |
Mean follow-up years |
||
|
MVP |
MVR |
MVP |
MVR |
||||||
|
Antunes 1990 [14] |
South Africa |
1976–1984 |
241 |
675 |
21.5 |
27.1 |
\ |
\ |
\ |
|
Brescia 2022 [15] |
USA |
1997–2018 |
80 |
100 |
\ |
\ |
\ |
\ |
5.0 ± 3.9 |
|
Cotrufo 1997 [17] |
Italy |
1981–1996 |
300 |
240 |
43 |
50 |
34/266 |
66/174 |
6.2± 1.7 |
|
Chen 2022 [16] |
China (Taiwan) |
2000–2013 |
467 |
467 |
56.8 |
56.7 |
219/248 |
206/261 |
5.9 ± 4.2 |
|
Duran 1991 [18] |
Saudi Arabia |
1988–1990 |
136 |
67 |
26.5 |
34 |
65/71 |
32/35 |
\ |
|
Duran 1994 [19] |
Saudi Arabia |
1988-1992 |
306 |
231 |
\ |
\ |
\ |
\ |
1.7 |
|
Fu 2020 [20] |
China |
2011–2019 |
529 |
529 |
54.5 |
54.6 |
147/382 |
147/382 |
4.1 |
|
Grossi 1998 [22] |
USA |
1980-1996 |
725 |
514 |
59.4 |
60.9 |
\ |
\ |
3.3 |
|
Geldenhuys 2012 [21] |
South Africa |
2000–2010 |
69 |
69 |
36.9 |
40.9 |
15/54 |
11/58 |
4.4 ± 3.0 |
|
Ho 2004 [23] |
Vietnam |
1992–2001 |
201 |
408 |
32.2 |
38.7 |
108/93 |
227/181 |
9 |
|
Jiao 2019 [24] |
China |
2011–2017 |
221 |
700 |
50 |
55.5 |
47/174 |
190/510 |
5.6 |
|
Kuwaki 2007 [27] |
Japan |
1981–2003 |
47 |
81 |
48 |
53 |
14/33 |
34/47 |
8 |
|
Kim 2010 [25] |
South Korea |
1997–2007 |
122 |
418 |
41.7 |
51 |
27/95 |
157/261 |
6.0 ± 3.3 |
|
Kim 2018 [26] |
South Korea |
1997–2005 |
294 |
1437 |
43.9 |
54 |
70/224 |
471/966 |
\ |
|
Krishna Moorthy 2018 [28] |
Malaysia |
1992–2015 |
336 |
83 |
12.3 |
13.8 |
133/203 |
36/47 |
3.9 |
|
Remenyi 2013 [29] |
New Zealand |
1990–2006 |
48 |
33 |
11.7 |
14.4 |
28/20 |
11/22 |
5 |
|
Russell 2017 [30] |
Australia |
2001–2013 |
119 |
1078 |
57.3 |
62 |
50/69 |
309/769 |
\ |
|
Talwar 2007 [31] |
India |
1995–2005 |
76 |
293 |
30.3 |
32.5 |
53/23 |
211/82 |
5.8 ± 3.4 |
|
Wang 2008 [32] |
China (Taiwan) |
1997–2005 |
33 |
59 |
49.7 |
58.1 |
12/21 |
20/39 |
3.0 ± 1.9 |
|
Yau 1999 [33] |
Canada |
1978–1995 |
142 |
431 |
42 |
57.9 |
21/121 |
88/343 |
5.7 ± 3.8 |
TABLE 2:
Prevalence of major risk factors in individual studies.
|
Study |
MECH (%) |
NYHA (II/III/IV%) |
AF (%) |
MR (%) |
MS (%) |
MR+MS (%) |
CS TVP (%) |
CS AVR (%) |
|||||||
|
MVP |
MVR |
MVP |
MVR |
MVP |
MVR |
MVP |
MVR |
MVP |
MVR |
MVP |
MVR |
MVP |
MVR |
||
|
Antunes 1990 [14] |
57.2 |
\ |
\ |
\ |
\ |
73.0 |
77.6 |
\ |
\ |
\ |
\ |
\ |
\ |
\ |
\ |
|
Brescia 2022 [15] |
\ |
\ |
\ |
\ |
\ |
\ |
\ |
\ |
\ |
\ |
\ |
\ |
\ |
\ |
\ |
|
Cotrufo 1997 [17] |
100 |
\ |
\ |
36.0 |
\ |
\ |
\ |
\ |
\ |
\ |
\ |
\ |
\ |
\ |
\ |
|
Chen 2022 [16] |
\ |
\ |
\ |
57.4 |
58.0 |
19.3 |
18.8 |
65.1 |
65.5 |
15.6 |
15.6 |
26.3 |
27.2 |
22.1 |
21.0 |
|
Duran 1991 [18] |
46.2 |
8.1/74.2/16.9 |
4.48/80.6/13.4 |
32.2 |
46.3 |
56.6 |
29.9 |
\ |
\ |
43.4 |
70.1 |
\ |
\ |
\ |
\ |
|
Duran 1994 [19] |
38.0 |
\ |
\ |
\ |
\ |
\ |
\ |
\ |
\ |
\ |
\ |
\ |
\ |
\ |
\ |
|
Fu 2020 [20] |
\ |
\ |
\ |
67.9 |
69.6 |
12.7 |
10.2 |
10.4 |
11.9 |
76.9 |
77.9 |
91.1 |
89.2 |
18.9 |
19.8 |
|
Grossi 1998 [22] |
100 |
\ |
\ |
\ |
\ |
49.1 |
27.8 |
\ |
\ |
\ |
\ |
\ |
\ |
\ |
\ |
|
Geldenhuys 2012 [21] |
91.3 |
\ |
\ |
29.0 |
39.0 |
39.0 |
13.0 |
4.3 |
10.7 |
39.1 |
71.0 |
5.8 |
2.9 |
\ |
\ |
|
Ho 2004 [23] |
99.0 |
79.6/17.9/1.5 |
83.8/15.2/0.5 |
36.8 |
60.3 |
37.4 |
12.7 |
30.3 |
59.1 |
32.3 |
28.2 |
28.9 |
32.4 |
100 |
100 |
|
Jiao 2019 [24] |
72.6 |
\ |
\ |
\ |
\ |
\ |
\ |
\ |
\ |
\ |
\ |
90.0 |
89.7 |
13.1 |
27.8 |
|
Kuwaki 2007 [27] |
81.5 |
-/-/12.7 |
-/-/18.1 |
\ |
\ |
8.5 |
6.2 |
83.0 |
54.3 |
8.5 |
39.5 |
17.0 |
40.7 |
100 |
100 |
|
Kim 2010 [25] |
100 |
\ |
\ |
72.9 |
82.5 |
26.2 |
66.0 |
67.2 |
14.8 |
8.2 |
19.1 |
\ |
\ |
\ |
\ |
|
Kim 2018 [26] |
78.9 |
\ |
\ |
56.8 |
75.2 |
61.6 |
17.5 |
24.1 |
46.4 |
14.3 |
36.1 |
33.6 |
43.2 |
17.3 |
34.3 |
|
Krishna Moorthy 2018 [28] |
83.1 |
II+III+IV: 70.5 |
II+III+IV: 66.3 |
\ |
\ |
93.8 |
90.4 |
0.9 |
4.8 |
5.4 |
4.8 |
31.5 |
14.4 |
\ |
\ |
|
Remenyi 2013 [29] |
100 |
2.79±0.6 |
2.81±0.7 |
6.0 |
21.0 |
86.0 |
87.9 |
8.0 |
0 |
6.0 |
12.1 |
\ |
\ |
\ |
\ |
|
Russell 2017 [30] |
100 |
III+IV: 42.9 |
II+III+IV: 66.3 |
26.1 |
48.9 |
74.8 |
24.7 |
6.7 |
34.2 |
18.5 |
36.1 |
\ |
\ |
\ |
\ |
|
Talwar 2007 [31] |
100 |
II+IV: 48.0 |
III+IV: 45.5 |
48.7 |
38.3 |
15.8 |
13.2 |
40.8 |
44.4 |
43.4 |
42.4 |
\ |
\ |
100 |
100 |
|
Wang 2008 [32] |
69.5 |
12.5/66.7/18.2 |
5.1/42.4/8.5 |
93.9 |
96.6 |
14.4 |
14.4 |
15.2 |
15.2 |
70.7 |
70.7 |
45.5 |
61.0 |
\ |
\ |
|
Yau 2000 [33] |
62.4 |
25/63.2/11.8 |
28.1/64.1/7.8 |
31.7 |
64.3 |
16.2 |
14.8 |
67.6 |
48.0 |
16.2 |
37.1 |
7.8 |
18.1 |
\ |
\ |
MVP: Mitral Valve Repair; MVR: Mitral Valve Replacement; MECH: Mechanical Valves; NYHA: New York Heart Association; AF: Atrial Fibrillation; MR: Mitral Regurgitation; MS: Mitral Stenosis; CS: Concomitant Surgery; TVP: Tricuspid Valve; AVR: Aortic Valve Replacement.
3.2. Early Mortality
There was no significant heterogeneity in statistics (heterogeneity test I² = 11.2%, P = 0.317), the fixed effect model could be directly used for meta-analysis, the merger effect was statistically significant. MVP was found to significantly reduce the risk of early mortality compared with MVR (OR, 0.63; 95% CI: 0.50-0.78; P < 0.001), as shown in (Figure 4 & Table 3). The funnel plot symmetry for assessing early mortality is shown in (Figure 5), with Begg's test (P > 0.05) and Egger's test (P > 0.05) indicating no evidence of publication bias.
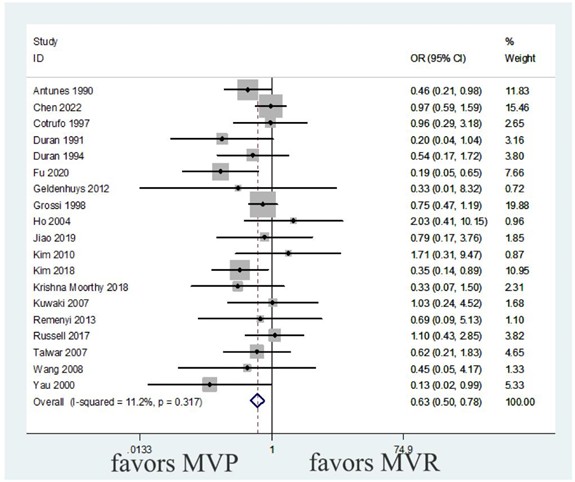
MVP: Mitral Valve Repair; MVR: Mitral Valve Replacement.
TABLE 3: Meta-analysis
of clinical outcomes of mitral valve repair and replacement.
|
Outcome |
Subgroup |
N |
Heterogeneity
test |
Z test |
OR (95%
CI) |
Publication
bias |
|||
|
|
|
|
PH |
I²(%) |
PZ |
Fixed
model |
Random
model |
PB |
PE |
|
Early
mortality |
|
19 |
0.317 |
11.2 |
< 0.001 |
0.63
(0.50–0.78) |
|
1.000 |
0.662 |
|
Long-term
mortality |
|
14 |
0.001 |
61.8 |
<
0.001 |
|
0.57
(0.42–0.77) |
0.743 |
0.118 |
|
Reoperation
rate |
|
|
0.048 |
44.6 |
<
0.001 |
|
2.73
(1.86–4.00) |
0.837 |
0.776 |
|
|
Before
2000 |
5 |
0.042 |
59.7 |
<
0.001 |
|
3.67
(1.98–6.78) |
0.221 |
0.041 |
|
|
2000–2010 |
5 |
0.090 |
50.3 |
0.040 |
|
2.16
(1.04–4.50) |
0.806 |
0.541 |
|
|
After 2010 |
2 |
0.286 |
12.3 |
0.222 |
|
1.81
(0.70–4.68) |
1.000 |
\ |
|
Valve
related events |
|
|
0.127 |
24.3 |
<
0.001 |
0.67
(0.57–0.79) |
|
|
|
|
|
Infective
endocarditis |
9 |
0.892 |
0 |
0.786 |
1.07
(0.66–1.73) |
|
0.076 |
0.035 |
|
|
Thromboembolism |
10 |
0.088 |
40.4 |
<
0.001 |
0.58
(0.46–0.74) |
|
1.000 |
0.208 |
|
|
Bleeding |
8 |
0.098 |
24.3 |
0.004 |
0.70
(0.55–0.89) |
|
0.536 |
0.058 |
|
Adverse
events |
|
|
0.804 |
0 |
0.013 |
0.76
(0.62–0.94) |
|
|
|
|
|
Heart
failure |
4 |
0.688 |
0 |
0.004 |
0.28
(0.12–0.67) |
|
0.734 |
0.363 |
|
|
Stroke |
5 |
0.933 |
0 |
0.503 |
0.83
(0.48–1.44) |
|
0.462 |
0.429 |
|
|
Fibrillation |
4 |
0.842 |
0 |
0.180 |
0.85
(0.66–1.08) |
|
0.734 |
0.808 |
N: Number; PH: P-value of the heterogeneity test; PZ: P-value of Z test; OR: Odds Ratio; 95% CI: 95% Confidence Interval; PB: P-value of Begg’s test; PE: P-value of Egger’s test.
3.3. Long-Term Mortality
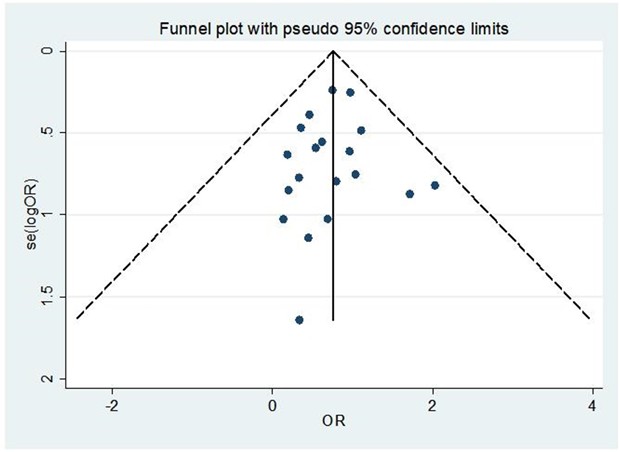
There was moderate heterogeneity in statistics (heterogeneity test I² = 61.8%, P = 0.001). Two studies were combined using HR instead of OR [15, 20]. The sensitivity analysis of the heterogeneity source was carried out, and the inclusion studies were eliminated one by one, the results were relatively stable (Supplementary Figure 1). Therefore, the random effect model is used to carry out the combined effect. MVP was found to significantly reduce the risk of long-term mortality compared with MVR (OR: 0.57, 95% CI: 0.42-0.77, P < 0.001), as shown in (Figure 6 and detailed in Table 3). The quantitative results of the Begg’s test (P > 0.05) and Egger’s test (P > 0.05), along with the funnel plot for long-term mortality shown in (Figure 7), suggest no evidence of publication bias.
MVP: Mitral Valve Repair; MVR: Mitral Valve Replacement.
3.4. Reoperation Rates
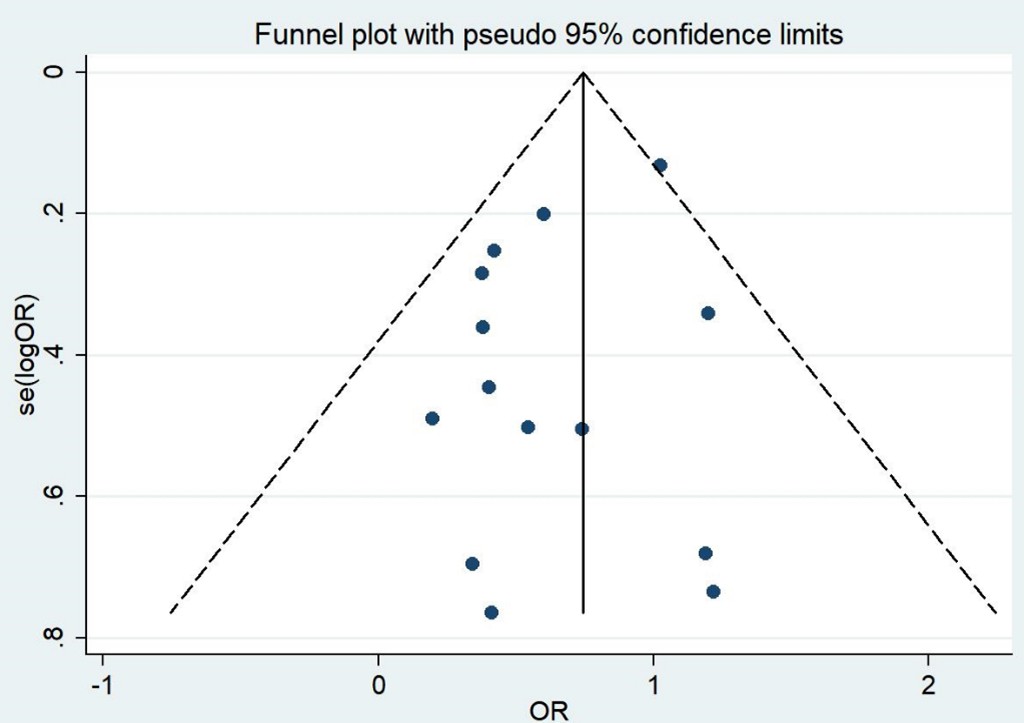
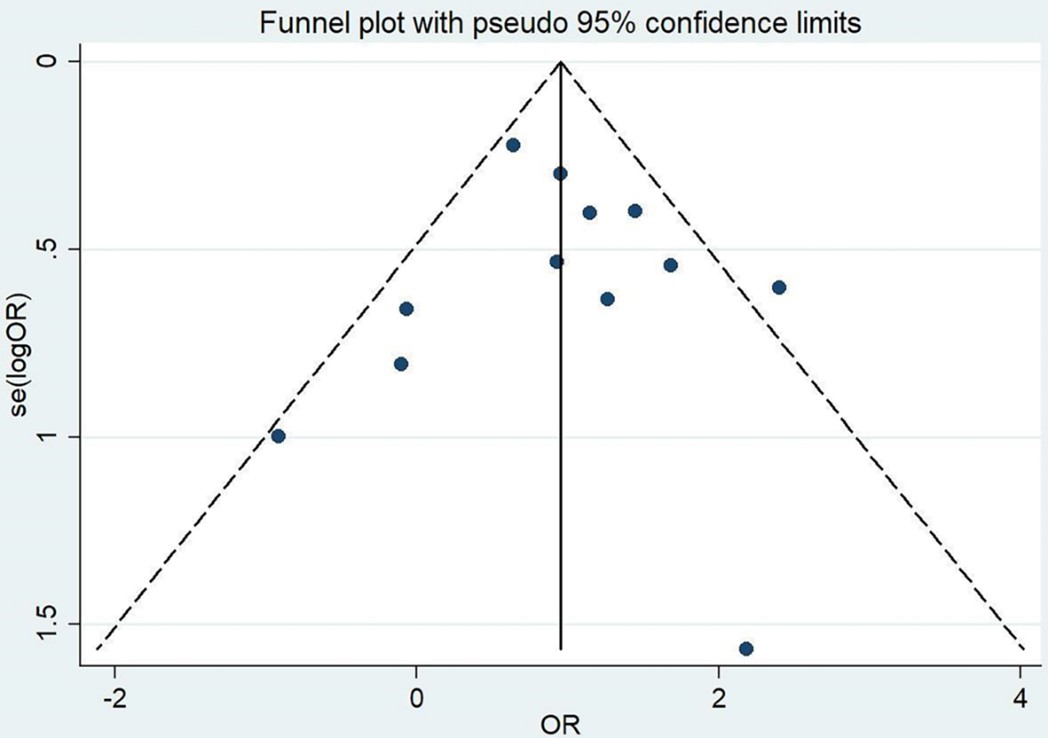
It showed mild heterogeneity (heterogeneity test I²=44.6%, P=0.048). Two studies combined HR instead of OR [20, 21]. The random effect model was employed with the year of surgery as a subgroup, excluding studies with a large span of years of surgery and retaining studies with a smaller span of years of surgery. Before 2000, MVP was associated with a high risk of reoperation (OR, 3.67; 95% CI: 1.98-6.78, P < 0.001). From 2000 to 2010, the risk of reoperation decreased compared to the previous period but remained relatively high overall (OR: 2.16, 95% CI: 1.04-4.50, P = 0.040). After 2010, the reoperation rate for MVP approached that for MVR (OR, 1.81; 95% CI: 0.70-4.68, P = 0.222), as shown in (Figure 8 & Table 3).
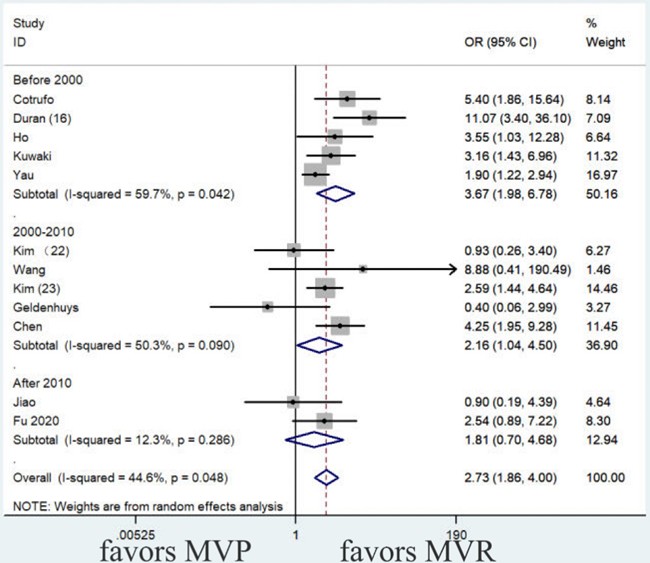
MVP: Mitral Valve Repair; MVR: Mitral Valve Replacement.
We used funnel plots, Begg’s test, and Egger’s test to evaluate publication bias. The funnel plot is symmetrical, as shown in (Figure 9). The results of the Begg’s test (P > 0.05) and Egger’s test (P > 0.05) did not indicate any publication bias.
3.5. Valve-Related Events
The heterogeneity test revealed no heterogeneity in any of the three subgroups (heterogeneity test I² = 24.3%, P = 0.127), which were combined using the fixed effect model. Compared with MVR, MVP reduced the risk of thromboembolism (OR: 0.58, 95% CI: 0.46-0.74, P < 0.001) and bleeding (OR: 0.70, 95% CI: 0.55-0.89, P = 0.004), but did not significantly affect the risk of infective endocarditis (OR: 1.07, 95% CI: 0.66-1.73, P = 0.786), as shown in (Figure 10 & Table 3).
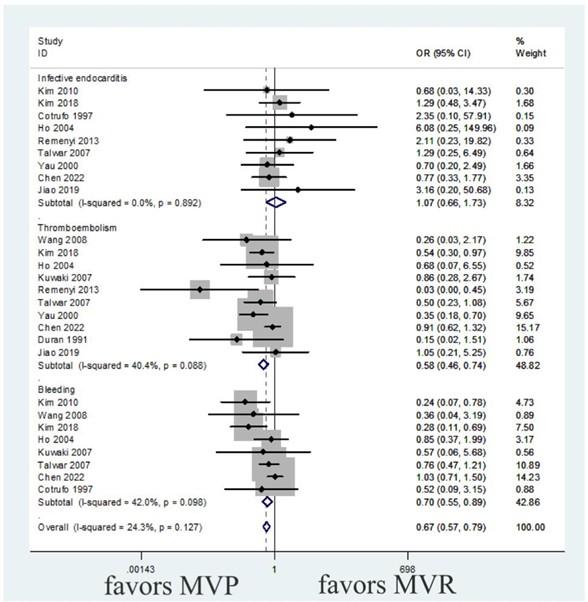
MVP: Mitral Valve Repair; MVR: Mitral Valve Replacement.
3.6. Adverse Events
The heterogeneity test revealed no heterogeneity in any of the three subgroups (heterogeneity test I² = 0.0%, P = 0.779). Using the fixed effect model for the meta-analysis, MVP was found to be more effective than MVR in reducing the risk of heart failure (OR: 0.28, 95% CI: 0.12-0.67, P = 0.004). However, there was no significant difference between the two groups in terms of stroke (P = 0.503) or atrial fibrillation (P = 0.180), as shown in (Figure 11 & Table 3).
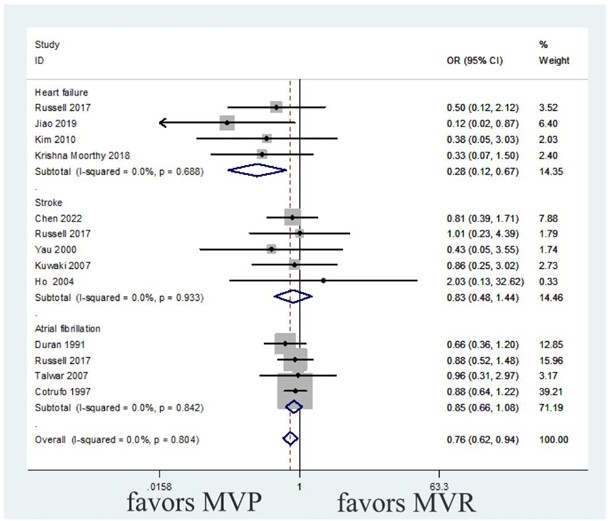
MVP: Mitral Valve Repair; MVR: Mitral Valve Replacement.
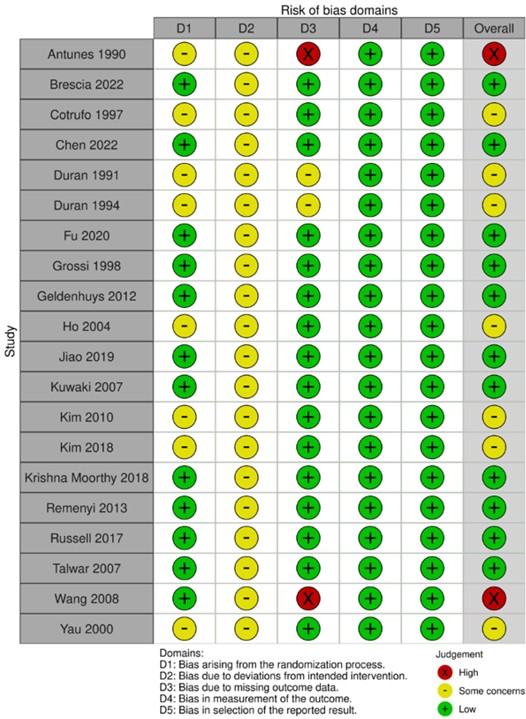
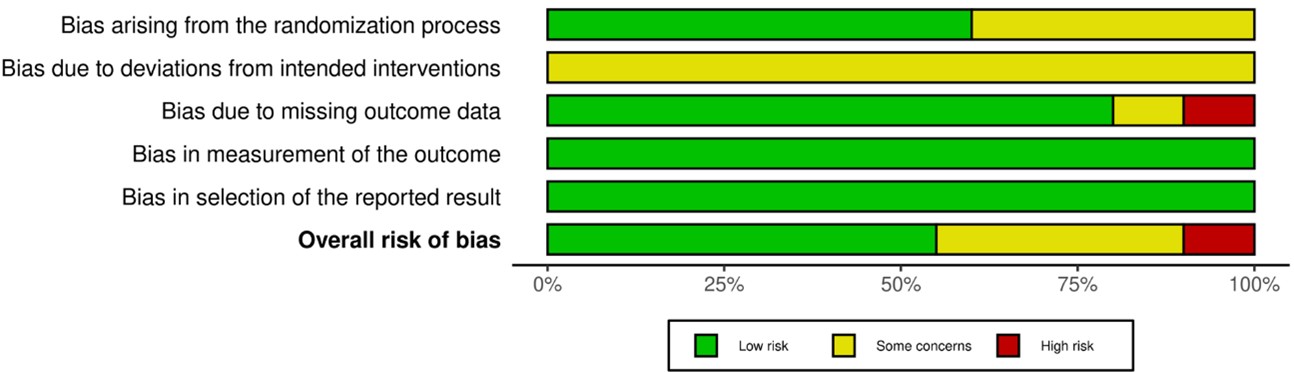
3.7. Risk of Bias
ROBINS-II is used for risk of bias assessment. Risk of bias for each of the studies are shown in (Figure 2). Summarise the risk of bias analysis of all studies, as shown in (Figure 3).
4. Discussion
In degenerative mitral valve disease, MVP is regarded as the gold standard for surgical treatment and has a lower early mortality than MVR [34, 35]. The main cause of early postoperative mortality in these patients was cardiac insufficiency. MVP usually avoids the need for resection of sub-valvular structures, preserves the integrity of the autologous valve, reduces cardiac damage, and minimizes the recovery time after surgery [24, 36]. It is also associated with a better left ventricular ejection fraction [32], reducing the incidence of events such as heart failure due to left heart dysfunction [24, 25, 30]. By the same principle, in rheumatic mitral valve disease, MVP also achieves superior early survival benefits compared with MVR [37]. This study found that rheumatic MVP significantly lowered early mortality relative to MVR (OR: 0.63), which is consistent with a previous study [10]. In addition, rheumatic MVP reduces early valve-related complications, including valve detachment and valve thrombosis, which likely contributes to the observed reduction in early mortality [20, 38, 39].
This study showed that rheumatic MVP had lower long-term mortality than MVR (OR, 0.57; 95% CI: 0.42-0.77; P < 0.001). MVP is advantageous in preserving left heart function, avoiding the long-term need for anticoagulant medication [26, 31, 38], and reducing the incidence of fatal long-term events. A previous study involving 2770 rheumatic MVP cases reported a 10-year long-term survival rate of 97.3% [40]. Furthermore, a 30-year follow-up showed that patients with rheumatic MVP had a long-term survival rate comparable to that of patients with degenerative disease [41]. In addition, MVP reduces the incidence of thromboembolic and bleeding events compared with prolonged anticoagulation therapy [31, 32, 42], which contributes to decreased long-term mortality. The present study also showed that MVP significantly reduced the risk of thromboembolism and bleeding, in agreement with the findings of a previous study [43].
The most controversial issue is reoperation. Because of significant changes in case selection and surgical techniques, it is not reasonable to analyze all studies together. Therefore, the studies were divided into three subgroups and analyzed using the year of surgery as a boundary. Studies prior to 2000 consistently reported a high risk of reoperation for MVP [17, 19, 23, 27, 33]. Degenerative mitral valve lesions are mainly characterized by localized pathological damage, whereas RHD affects the entire mitral valve apparatus, including the annulus, valve body, sub-valvular tendon cords, and papillary muscles, which may be damaged and altered to varying degrees [44]. It is generally accepted that the degree of pathologic damage to the rheumatic mitral valve affects the application of the repair technique [45, 46]. Furthermore, Yau et al. [17, 33] showed that factors such as significant damage to the mitral valve tissue contribute to a high rate of repair failure. In patients with RHD, MVP combined with aortic valve replacement, compared with MVR combined with aortic valve replacement, has been reported by Ho et al. [27] to have a significantly higher reoperation rate. This is mainly due to the progression of mitral valve disease and the deterioration of the bioprosthetic aortic valve [10, 47]. Therefore, MVP should be limited to repairing lesions with better durability. The reoperation rates for MVP are also influenced by age [48, 49]; and Duran et al. [19] found a higher failure rate among patients under 20 years of age who opted for MVP than among older patients with rheumatic MVP. Therefore, studies before 2000 may have had higher reoperation rates possibly because of inappropriate case selection.
The period from 2000 to 2010 was transitional, and studies have shown that the risk of reoperation has trended downward, with large variations in reoperation rates between institutions. Chen and Wang [16, 32] showed that MVP had a higher reoperation rate than MVR. They identified factors such as prior percutaneous transvenous mitral commissurotomy, complex valve pathology, and other factors associated with higher rates of mitral valve reoperation. Chen also concluded that it is important to have an experienced surgeon evaluate mitral calcification and rheumatologic activity in early stage patients with rheumatic mitral regurgitation. Kim et al. [25, 26] showed that MVP has a favorable outcome compared to mechanical valve replacement for RHD in selected patients. MVP has good durability compared to mechanical valve replacement for RHD, and there was no significant difference in the incidence of reoperation between the two approaches. In this center, MVP is mainly performed by junctional dissection, and valve replacement is more frequently chosen when severe fibrosis or calcification affects the mobility of the anterior leaflet or involves sub-valvular structures. This suggests that rational patient selection and advancements in surgical technique have led to improved reoperation rates for rheumatic MVP [25, 26, 50].
Since 2010, two studies have shown that there is no longer a significant difference in reoperation rates between MVP and MVR [20, 24]. The current understanding of rheumatic mitral valve disease is improving and MVP techniques have also improved significantly. Recently, Nguyen et al. suggested many reasons for reoperation following rheumatic MVP, such as suture dehiscence, shortened tendon cable rupture, incorrect selection of the size of the shaped ring, and lack of valve tissue. Therefore, it is necessary for the operator to have a good understanding of the severity of the valve lesion, choose the appropriate repair method, and demonstrate successful repair experience [51].
Important advances in rheumatic MVP have been made in recent years. Some researchers have proposed a three-part clinicopathologic triage of rheumatic mitral valves [52], comprising leaflet thickening, sub-valvular structure thickening, and mixed-type. This typing method can help surgeons determine a patient's condition more accurately and thus develop a more personalized treatment plan [53]. Building on this foundation, researchers have developed a standardized procedure for rheumatic MVP surgery--the "SCOR" procedure--specifically designed to repair the fused commissure [20, 52, 54]. The four procedures are shaving junctional fibro-plaques, checking the natural junctional area, performing junctional commissurotomy, and relaxing adhesions in the sub-valvular structures. Significant clinical results have been obtained in practice. The difficulty of the procedure has been reduced, which significantly decreases the reoperation rate [20, 24]. This has greatly contributed to the development of rheumatic MVP. Therefore, we believe that rational case selection and improvement in repair techniques are key initiatives that have led to optimal results in rheumatic MVP.
5. Conclusion
In patients with RHD, MVP reduces early and long-term mortality, thromboembolism, bleeding, and heart failure compared with replacement. With proper case selection and improved repair techniques, the reoperation rates have significantly decreased, especially after 2010. Currently, there is no significant difference in the reoperation rates between the two procedures. In conclusion, MVP is the preferred treatment of choice for selected patients with RHD.
Limitations
This meta-analysis has several limitations. First, the studies cited in this review were primarily retrospective cohort studies and lacked randomized controlled trials. Second, the sample sizes of some studies were too small, which unavoidably increased selection bias in the analysis. Third, the studies were conducted in different countries, which may be heterogeneous, and several studies did not report baseline parameters for patients, making it impossible to obtain level data for all patients.
Author Contributions
Chuan Yuan: Conceptualization, data curation, methodology, writing - original draft. Kejun Liu: Conceptualization, Data curation, Methodology, Writing - original draft. Yi Liu: Conceptualization, data curation, methodology, writing - original draft. Sihao Zhou: Conceptualization, data curation, methodology, writing - original draft. XueShan Huang: Conceptualization, methodology. Yingmeng Wu: Formal analysis, Validation. Hongyu Ye: Formal analysis, Validation. Yi Liang: Methodology, supervision, writing - review, and editing. Weizhao Huang: Methodology, supervision, writing - review, and editing. All authors have contributed.
Acknowledgements
Not applicable.
Funding
None.
Conflicts of Interest
None.
Assistance with the Study
None.
Presentation
None.
Ethical Approval
Not applicable.
Provenance and Peer Review
Not commissioned, externally peer-reviewed.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
REFERENCES
[1] Sean Coffey, Ross Roberts-Thomson, Alex Brown, et al.
“Global epidemiology of valvular heart disease.” Nat Rev Cardiol, vol.
18, no. 12, pp. 853-864, 2021. View at: Publisher Site | PubMed
[2] Eloi Marijon, Mariana Mirabel, David S Celermajer, et
al. “Rheumatic heart disease.” Lancet, vol. 379, no. 9819, pp. 953-964,
2012. View at: Publisher Site | PubMed
[3] C N Manjunath, P Srinivas, K S Ravindranath, et al.
“Incidence and patterns of valvular heart disease in a tertiary care
high-volume cardiac center: a single center experience.” Indian Heart J,
vol. 66, no. 3, pp. 320-326, 2014. View at: Publisher Site | PubMed
[4] Catherine M Otto, Rick A Nishimura, Robert O Bonow, et
al. “2020 ACC/AHA Guideline for the Management of Patients With Valvular Heart
Disease: Executive Summary: A Report of the American College of
Cardiology/American Heart Association Joint Committee on Clinical Practice
Guidelines.” Circulation, vol. 143, no. 5, pp. e35-e71, 2021. View at: Publisher Site | PubMed
[5] Damien J LaPar, Gorav Ailawadi, James M Isbell, et al.
“Mitral valve repair rates correlate with surgeon and institutional
experience.” J Thorac Cardiovasc Surg, vol. 148, no. 3, pp. 995-1004,
2014. View at: Publisher Site | PubMed
[6] Helmut Baumgartner, Volkmar Falk, Jeroen J Bax, et al.
“2017 ESC/EACTS Guidelines for the management of valvular heart disease.” Eur
Heart J, vol. 38, no. 36, pp. 2739-2791, 2017. View at: Publisher Site | PubMed
[7] Christina M Vassileva, Christian McNeely, John
Spertus, et al. “Hospital volume, mitral repair rates, and mortality in mitral
valve surgery in the elderly: an analysis of US hospitals treating Medicare
fee-for-service patients.” J Thorac Cardiovasc Surg, vol. 149, no. 3,
pp. 762-768.e1, 2015. View at: Publisher Site | PubMed
[8] Rakesh M Suri, Jean-Louis Vanoverschelde, Francesco
Grigioni, et al. “Association between early surgical intervention vs watchful
waiting and outcomes for mitral regurgitation due to flail mitral valve
leaflets.” JAMA, vol. 310, no. 6, pp. 609-616, 2013. View at: Publisher Site | PubMed
[9] Somchai Waikittipong “Mitral valve repair for
rheumatic mitral regurgitation: Mid-term results.” Asian Cardiovasc Thorac
Ann, vol. 23, no. 6, pp. 658-664, 2015. View at: Publisher Site | PubMed
[10]
Alok Saurav,
Venkata Mahesh Alla, Manu Kaushik, et al. “Outcomes of mitral valve repair
compared with replacement in patients undergoing concomitant aortic valve
surgery: a meta-analysis of observational studies.” Eur J Cardiothorac Surg,
vol. 48, no. 3, pp. 347-353, 2015. View at: Publisher Site | PubMed
[11]
Brian Hutton,
Georgia Salanti, Deborah M Caldwell, et al. “The PRISMA extension statement for
reporting of systematic reviews incorporating network meta-analyses of health
care interventions: checklist and explanations.” Ann Intern Med, vol.
162, no. 11, pp. 777-784, 2015. View at: Publisher Site | PubMed
[12]
Beverley J
Shea, Barnaby C Reeves, George Wells, et al. “AMSTAR 2: a critical appraisal
tool for systematic reviews that include randomised or non-randomised studies
of healthcare interventions, or both.” BMJ, vol. 358, pp. j4008, 2017. View at: Publisher Site | PubMed
[13] Jonathan A C Sterne, Jelena Savović, Matthew J
Page, et al. “RoB 2: a revised tool for assessing risk of bias in
randomised trials.” BMJ, vol. 366, pp. l4898, 2019. View at: Publisher Site | PubMed
[14]
M J Antunes
“Mitral valvuloplasty, a better alternative. Comparative study between valve
reconstruction and replacement for rheumatic mitral valve disease.” Eur J
Cardiothorac Surg, vol. 4, no. 5, pp. 257-264, 1990. View at: Publisher Site | PubMed
[15]
Alexander A
Brescia, Tessa M F Watt, Shannon L Murray, et al. “Rheumatic mitral valve
repair or replacement in the valve-in-valve era.” J Thorac Cardiovasc Surg,
vol. 163, no. 2, pp. 591-602.e1, 2022. View at: Publisher Site | PubMed
[16]
Shao-Wei Chen,
Cheng-Yu Chen, Victor Chien-Chia Wu, et al. “Mitral valve repair versus
replacement in patients with rheumatic heart disease.” J Thorac Cardiovasc
Surg, vol. 164, no. 1, pp. 57-67.e11, 2022. View at: Publisher Site | PubMed
[17]
M Cotrufo, A
Renzulli, N Vitale, et al. “Long-term follow-up of open commissurotomy versus
bileaflet valve replacement for rheumatic mitral stenosis.” Eur J
Cardiothorac Surg, vol. 12, no. 3, pp. 335-340, 1997. View at: Publisher Site | PubMed
[18]
C M Duran, B
Gometza, S Balasundaram, et al. “A feasibility study of valve repair in
rheumatic mitral regurgitation.” Eur Heart J, vol. 12 Suppl B, pp.
34-38, 1991. View at: Publisher Site | PubMed
[19]
C M Duran, B
Gometza, E Saad “Valve repair in rheumatic mitral disease: an unsolved
problem.” J Card Surg, vol. 9, no. 2 Suppl, pp. 282-285, 1994. View at: Publisher Site | PubMed
[20]
Jintao Fu, Yan
Li, Haibo Zhang, et al. “Outcomes of mitral valve repair compared with
replacement for patients with rheumatic heart disease.” J Thorac Cardiovasc
Surg, vol. 162, no. 1, pp. 72-82.e7, 2021. View at: Publisher Site | PubMed
[21]
Agneta
Geldenhuys, Jithan J Koshy, Paul A Human, et al. “Rheumatic mitral repair
versus replacement in a threshold country: the impact of commissural fusion.” J
Heart Valve Dis, vol. 21, no. 4, pp. 424-432, 2012. View at: PubMed
[22]
E A Grossi, A
C Galloway, J S Miller, et al. “Valve repair versus replacement for mitral
insufficiency: when is a mechanical valve still indicated?” J Thorac
Cardiovasc Surg, vol. 115, no. 2, pp. 389-396, 1998. View at: Publisher Site | PubMed
[23]
Huynh-Quang
Tri Ho, Van-Phan Nguyen, Kim-Phuong Phan, et al. “Mitral valve repair with
aortic valve replacement in rheumatic heart disease.” Asian Cardiovasc
Thorac Ann, vol. 12, no. 4, pp. 341-345, 2004. View at: Publisher Site | PubMed
[24]
Yuqing Jiao,
Tiange Luo, Haibo Zhang, et al. “Repair versus replacement of mitral valves in
cases of severe rheumatic mitral stenosis: mid-term clinical outcomes.” J
Thorac Dis, vol. 11, no. 9, pp. 3951-3961, 2019. View at: Publisher Site | PubMed
[25]
Joon Bum Kim,
Hee Jung Kim, Duk Hwan Moon, et al. “Long-term outcomes after surgery for
rheumatic mitral valve disease: valve repair versus mechanical valve
replacement.” Eur J Cardiothorac Surg, vol. 37, no. 5, pp. 1039-1046,
2010. View at: Publisher Site | PubMed
[26]
Wan Kee Kim,
Ho Jin Kim, Joon Bum Kim, et al. “Clinical outcomes in 1731 patients undergoing
mitral valve surgery for rheumatic valve disease.” Heart, vol. 104, no.
10, pp. 841-848, 2018. View at: Publisher Site | PubMed
[27]
Kenji Kuwaki,
Nobuyoshi Kawaharada, Kiyofumi Morishita, et al. “Mitral valve repair versus
replacement in simultaneous mitral and aortic valve surgery for rheumatic
disease.” Ann Thorac Surg, vol. 83, no. 2, pp. 558-563, 2007. View at: Publisher Site | PubMed
[28]
Paneer Selvam
Krishna Moorthy, Sivakumar Sivalingam, Jeswant Dillon, et al. “Is it worth
repairing rheumatic mitral valve disease in children? Long-term outcomes of an
aggressive approach to rheumatic mitral valve repair compared to replacement in
young patients.” Interact Cardiovasc Thorac Surg, vol. 28, no. 2, pp.
191-198, 2019. View at: Publisher Site | PubMed
[29]
Bo Remenyi,
Rachel Webb, Tom Gentles, et al. “Improved long-term survival for rheumatic
mitral valve repair compared to replacement in the young.” World J Pediatr
Congenit Heart Surg, vol. 4, no. 2, pp. 155-164, 2013. View at: Publisher Site | PubMed
[30]
E Anne
Russell, Warren F Walsh, Christopher M Reid, et al. “Outcomes after mitral
valve surgery for rheumatic heart disease.” Heart Asia, vol. 9, no. 2,
pp. e010916, 2017. View at: Publisher Site | PubMed
[31]
Sachin Talwar,
Ankit Mathur, Shiv Kumar Choudhary, et al. “Aortic valve replacement with
mitral valve repair compared with combined aortic and mitral valve
replacement.” Ann Thorac Surg, vol. 84, no. 4, pp. 1219-1225, 2007. View at: Publisher Site | PubMed
[32]
Yao-Chang
Wang, Feng-Chun Tsai, Jaw-Ji Chu, et al. “Midterm outcomes of rheumatic mitral
repair versus replacement.” Int Heart J, vol. 49, no. 5, pp. 565-576,
2008. View at: Publisher Site | PubMed
[33]
T M Yau, Y A
El-Ghoneimi, S Armstrong, et al. “Mitral valve repair and replacement for
rheumatic disease.” J Thorac Cardiovasc Surg, vol. 119, no. 1, pp.
53-60, 2000. View at: Publisher Site | PubMed
[34]
Michele De
Bonis, Ottavio Alfieri, Malcolm Dalrymple-Hay, et al. “Mitral Valve Repair in
Degenerative Mitral Regurgitation: State of the Art.” Prog Cardiovasc Dis,
vol. 60, no. 3, pp. 386-393, 2017. View at: Publisher Site | PubMed
[35]
Gonçalo F
Coutinho, Manuel J Antunes “Mitral valve repair for degenerative mitral valve
disease: surgical approach, patient selection and long-term outcomes.” Heart,
vol. 103, no. 21, pp. 1663-1669, 2017. View at: Publisher Site | PubMed
[36]
Gokhan Lafci,
Kerim Cagli, Omer Faruk Cicek, et al. “Papillary muscle repositioning as a
subvalvular apparatus preservation technique in mitral stenosis patients with
normal left ventricular systolic
function.” Tex Heart Inst J, vol. 41, no. 1, pp. 33-39, 2014. View at: Publisher Site | PubMed
[37]
Jeffrey
Shuhaiber, Robert J Anderson “Meta-analysis of clinical outcomes following
surgical mitral valve repair or replacement.” Eur J Cardiothorac Surg,
vol. 31, no. 2, pp. 267-275, 2007. View at: Publisher Site | PubMed
[38]
Zhenhua Wang,
Chenghui Zhou, Haiyong Gu, et al. “Mitral valve repair versus replacement in
patients with rheumatic heart disease.” J Heart Valve Dis, vol. 22, no.
3, pp. 333-339, 2013. View
at: PubMed
[39]
E M Lee, L M
Shapiro, F C Wells “Importance of subvalvular preservation and early operation
in mitral valve surgery.” Circulation, vol. 94, no. 9, pp. 2117-2123,
1996. View at: Publisher Site | PubMed
[40]
Jin-Tao Fu,
Mohammad Sharif Popal, Hai-Bo Zhang, et al. “A meta-analysis of late outcomes
of mitral valve repair in patients with rheumatic heart disease.” J Thorac
Dis, vol. 9, no. 11, pp. 4366-4375, 2017. View at: Publisher Site | PubMed
[41]
Daniel J
DiBardino, Andrew W ElBardissi, R Scott McClure, et al. “Four decades of
experience with mitral valve repair: analysis of differential indications,
technical evolution, and long-term outcome.” J Thorac Cardiovasc Surg,
vol. 139, no. 1, pp. 76-84, 2010. View at: Publisher Site | PubMed
[42]
M Ibrahim, H
O'Kane, J Cleland, et al. “The St. Jude Medical prosthesis. A thirteen-year
experience.” J Thorac Cardiovasc Surg, vol. 108, no. 2, pp. 221-230,
1994. View at: PubMed
[43]
Yefan Jiang,
Chen Wang, Geng Li, et al. “Clinical outcomes following surgical mitral valve
repair or replacement in patients with rheumatic heart disease: a
meta-analysis.” Ann Transl Med, vol. 9, no. 3, pp. 204, 2021. View at: Publisher Site | PubMed
[44]
Charles A
Yankah, Henryk Siniawski, Christine Detschades, et al. “Rheumatic mitral valve
repair: 22-year clinical results.” J Heart Valve Dis, vol. 20, no. 3,
pp. 257-264, 2011. View at: PubMed
[45]
A Deloche, V A
Jebara, J Y Relland, et al. “Valve repair with Carpentier techniques. The
second decade.” J Thorac Cardiovasc Surg, vol. 99, no. 6, pp. 990-1002,
1990. View at: PubMed
[46]
Robert O
Bonow, Blase A Carabello, Chatterjee Kanu, et al. “ACC/AHA 2006 guidelines for
the management of patients with valvular heart disease: a report of the
American College of Cardiology/American Heart Association Task Force on
Practice Guidelines (writing committee to revise the 1998 Guidelines for the
Management of Patients With Valvular Heart Disease): developed in collaboration
with the Society of Cardiovascular Anesthesiologists: endorsed by the Society
for Cardiovascular Angiography and Interventions and the Society of Thoracic
Surgeons.” Circulation, vol. 114, no. 5, pp. e84-e231, 2006. View at: Publisher Site | PubMed
[47]
Masaki
Hamamoto, Ko Bando, Junjiro Kobayashi, et al. “Durability and outcome of aortic
valve replacement with mitral valve repair versus double valve replacement.” Ann
Thorac Surg, vol. 75, no. 1, pp. 28-34, 2003. View at: Publisher Site | PubMed
[48]
S K Choudhary,
S Talwar, B Dubey, et al. “Mitral valve repair in a predominantly rheumatic
population. Long-term results.” Tex Heart Inst J, vol. 28, no. 1, pp.
8-15, 2001. View at: PubMed
[49]
Charles A
Yankah, Henryk Siniawski, Christine Detschades, et al. “Rheumatic mitral valve
repair: 22-year clinical results.” J Heart Valve Dis, vol. 20, no. 3,
pp. 257-264, 2011. View at: PubMed
[50]
Ali Fedakar,
Ahmet Sasmazel, Onursal Bugra, et al. “Results of mitral valve repair in
rheumatic mitral lesions.” Heart Surg Forum, vol. 13, no. 2, pp.
E86-E90, 2010. View at: Publisher Site | PubMed
[51]
Tiange Luo, Xu
Meng “Clinico-pathological classification of rheumatic mitral valve damage and
surgical strategy.” J Thorac Dis, vol. 13, no. 5, pp. 2933-2941, 2021. View at: Publisher Site | PubMed
[52]
Tiange Luo, Xu
Meng, Zhiguo Yan, et al. “Commissuroplasty as a Main Operative Technique in
Rheumatic Mitral Valve Repair: Surgical Experiences and Mid-Term Results.” Heart
Lung Circ, vol. 29, no. 6, pp. 940-948, 2020. View at: Publisher Site | PubMed
[53]
Jeswant Dillon, Mohd Azhari Yakub, Mohd Nazeri
Nordin, et al. “Leaflet extension in rheumatic mitral valve reconstruction.” Eur
J Cardiothorac Surg, vol. 44, no. 4, pp. 682-689. View at: Publisher Site | PubMed
